Copper-catalyzed asymmetric [4+1] annulation of yne–allylic esters with pyrazolones
-
* Corresponding author.
E-mail address: hao.xu@ccnu.edu.cn (H. Xu).
1 These authors contributed equally to this work.
Citation:
Guang Xu, Cuiju Zhu, Xiang Li, Kexin Zhu, Hao Xu. Copper-catalyzed asymmetric [4+1] annulation of yne–allylic esters with pyrazolones[J]. Chinese Chemical Letters,
;2025, 36(4): 110114.
doi:
10.1016/j.cclet.2024.110114

Point-of-care testing (POCT) technologies have attracted much attention owing to their potential to avoid complex laboratory operations and enable routine health monitoring [1-4]. With the current demand of personalized healthcare and home diagnosis, it is desired to develop POCT devices with the characteristics of simple operation, effective signal amplification, and portability for biomarker detection [5-8]. Miniaturized POCT devices have been recently developed, where electrochemistry [9,10], fluorescence [11,12], and microfluidics [13,14] were widely used owing to their low cost and rapid diagnosis. However, these methods require extra devices such as an electrochemical workstation, excitation light source, or injection pump, thus increasing the operation difficulty. Thus, they cannot satisfy the demand of easy-to-use devices for home diagnosis. Colorimetric assay can provide a power-free and straightforward readout signal by simple color changes, thus offering a solution for easy-to-use POCT devices [5,15,16].
Porous materials have been extensively developed as substrates for colorimetric assay, as their large specific surface area can provide abundant sites for probe molecule immobilization and analyte enrichment [17,18]. In recent years, TiO2 nanotube arrays (TiNTs) have attracted much interest ranging from drug delivery to biosensing owing to their intrinsic photocatalytic activity, high surface-to-volume ratio, and perpendicular structure to the substrate surface, and the presence of functional groups such as OH group on their surface [19-25]. Because TiNTs prepared by electrochemical anodization are readily adjustable in morphology (pore sizes, and aspect ratio), optical property, and surface charge, TiNTs can provide a flexible scaffold to load biomolecules or indicators for biosensing and optical detection [26-30]. Furthermore, amphiphilic nanotubes can be designed by utilizing the photocatalytic activity of TiO2 [31,32], thus allowing distinct regions in one substrate and expanding the functionality of TiNTs.
In this study, we designed hydrophilic patterns on TiNTs as nanoreactors for a highly sensitive naked-eye colorimetric assay. The fabrication procedure is shown in Fig. 1. In this design, TiNTs with a high aspect ratio and perpendicular arrays were grown with an electrochemical anodization and then removed from a Ti substrate to obtain a freestanding membrane. Then, the surface wettability of TiNTs was adjusted by coating a layer of hydrophobic molecules. By utilizing the photocatalytic activity of TiO2, hydrophilic patterns with a hydrophobic surrounding were fabricated on TiNTs via simple UV irradiation with aid of a mask. These small-sized hydrophilic compartments were then used as nanoreactors for colorimetric assay. As a proof-of-concept, alkaline phosphatase (ALP), an essential enzyme that relates to the dephosphorization of important life processes, was used as the model analyte to demonstrate the use of the proposed sensing platform. The detection mechanism is shown in Fig. 1. Hydrophilic nanoreactors were functionalized and loaded with indicators zincon-Zn(Ⅱ) chelates (ZC-Zn(Ⅱ)). As an enzyme substrate of ALP, pyrophosphate (PPi) can be hydrolyzed by ALP to phosphate ions (Pi). Moreover, the affinity of Zn(Ⅱ) to zincon is stronger than Pi and weaker than PPi. Therefore, the presence of PPi can trigger the release of red-colored zincon by competitive coordination of PPi-Zn(Ⅱ), while blue-colored ZC-Zn(Ⅱ) is insusceptible in the presence of Pi. Based on the distinct color responses, the proposed platform can provide a straightforward readout signal for ALP colorimetric assay.
TiNTs used in this study were prepared by electrochemical anodization in a NH4F-containing organic electrolyte. Scanning electron microscopy (SEM) images in Figs. 2A and B show that the as-prepared TiO2 nanotubes have a uniform porous nanostructure with a diameter of 150 ± 20 nm and length of ~33 µm, and TiNTs are composed of numerous perpendicular nanotubes with closed tube bottom (Fig. S1 in Supporting information). The specific Brunauer-Emmett-Teller (BET) surface areas of TiO2 NTs was determined as 47.459 m2/g (Fig. S2 in Supporting information), indicating the high specific surface area of TiNTs. To achieve high chemical stability, the TiNTs were annealed at 450 ℃ in air for 2 h. As most of the contamination inside TiNTs can be burnt off by high-temperature calcination, the color of TiNTs transferred to a pale yellow (Fig. 2C inset), which can offer a low background for naked-eye colorimetric assay. The X-ray diffraction (XRD) pattern in Fig. 2C shows specific diffraction peaks of anatase after the annealing treatment, suggesting the successful transfer from amorphous TiO2 to anatase. Compared with amorphous TiO2, anatase has a higher chemical stability and photocatalytic activity, which is beneficial for the fabrication of hydrophilic reactors on TiNTs. The colorimetric performance of TiNTs was then evaluated by adding the same volume of red-colored indicator (zincon, zinc metal-organic complex) to the glass slide, filter paper, and TiNTs. As expected, a much higher red-color intensity can be observed from TiNTs than other substrates (Fig. 2D). The large surface area of TiNTs offers a plenty of available sites for indicator attachment, while the nano test tube structure of TiNTs with a high aspect ratio can provide a long observation length combined with a limited volume. Therefore, TiNTs can amplify the optical signal and thus exhibit an excellent colorimetric performance.
By utilizing the intrinsic photocatalysis activity of TiO2, distinct hydrophilic/hydrophobic regions were fabricated on TiNTs to form small-sized nanoreactors to further improve the colorimetric performance. As pristine TiNTs showed a hydrophilic surface (Fig. 3A), a hydrophobic monolayer, octadecylphosphonic acid (ODPA), was coated on TiNTs, and the surface was converted to hydrophobic with a water contact angle of 140.2° (Fig. 3B). With abundant surface active sites in anodized TiNTs, hydrophobic ODPA was modified onto the nanotube via affinity interaction between the Ti-OH and phosphonic acid [33]. Then, the wettability of TiNTs was further adjusted by the decomposition of hydrophobic chains induced by the photocatalysis of TiO2 under UV irradiation. As shown in Fig. 3C, the water contact angle decreased to <5° after being exposed to UV light for a short time, suggesting the full degradation of hydrophobic chains on TiNTs. According to the abovementioned results, patterned hydrophilic regions were formed on hydrophobic TiNTs by covering a mask with open areas. Fig. 3D shows the digital image of patterned hydrophilic regions with a diameter of 1.0 mm on TiNTs. Because of the coverage of a patterned mask, photodegradation of hydrophobic chains is confined to areas exposed to UV light, while the hydrophobic chains in shadowed areas are reserved. With the hydrophobic surrounding, the blue indicators are well held in hydrophilic regions without spreading. Fig. 3E shows the cross-section fluorescent image of sodium fluorescein loaded hydrophilic compartments. With the limited transparent depth of UV light, the thickness of hydrophilic layer was ~24 µm, while the bottom of nanotubes exhibited a dark color without wetting by sodium fluorescein. Therefore, the volume of one hydrophilic compartment was calculated as 18.8 nL. These hydrophilic compartments with such a tight volume acted as nanoreactors, which can limit the indicators and analyte in a confined void, enabling the colorimetric assay with a high sensitivity.
To demonstrate the use of TiNTs for specific and sensitive analysis of disease-related biomarkers, we studied the detection of ALP using the proposed TiNTs in a colorimetric assay. First, the TiNTs were coated with a layer of poly(diallyldimethylammonium chloride) (PDDA), and thus abundant binding sites can be provided by the amino groups in PDDA for the grafting of negatively charged indicators via electrostatic interaction. The successful decoration of PDDA was confirmed by the positive shift of zeta potential from −24.7 mV to 33.9 mV (Fig. S3 in Supporting information). Then, blue-colored zincon-Zn(Ⅱ) chelates (ZC-Zn(Ⅱ)) as indicators were loaded by immersing bare TiNTs into a solution containing ZC-Zn(Ⅱ) after an incubation time of 35 min (Fig. S4 in Supporting information), and the substrate showed a blue color in hydrophilic nanoreactors (Fig. 3D). This step was then confirmed by X-ray photoelectron spectroscopy (XPS). As shown in Fig. 4A, the peaks of Zn 2p and N 1s appeared after the loading of ZC-Zn(Ⅱ), while decreased intensities of Ti 2p and O 1s peaks can be attributed to the coverage of ZC-Zn(Ⅱ) on the surface of TiO2. As the ALP activity is evaluated by its hydrolysis towards PPi, the response of ZC-Zn(Ⅱ) to PPi is crucial for colorimetric assay. As shown in Fig. 4B and Fig. S5 (Supporting information), the XPS peaks of Zn 2p showed a distinct shift to a higher energy in the presence of PPi. Because of the higher affinity between PPi and Zn2+ ions, ZC-Zn(Ⅱ) were substituted by the competitive coordination of PPi-Zn(Ⅱ) framework, thus resulting in a higher binding energy of Zn 2p. Effect of PPi concentrations to the color change of ZC-Zn(Ⅱ) chelate loaded TiNTs (ZZ/TiNTs) was further evaluated [34,35]. To provide a more accurate quantification of color variation, distribution of the red parameter of the Red-Green-Blue (RGB) system was analyzed using image J software, and the color variation of ZZ/TiNTs was quantified from the intensity difference of red parameter (ΔIred, calculated as I − I0, where I0 and I represent the intensities of red parameter of substrate before and after testing). As shown in Fig. 4C, ΔIred of ZZ/TiNTs continuously increased with increasing PPi concentration, while ΔIred became stable when the PPi concentration was more than 400 µmol/L. Hence, the PPi concentration in colorimetric assay was selected as 400 µmol/L, because excessive PPi will consume ALP and reduce the sensitivity of detection. As PPi can be hydrolyzed by ALP to Pi (H2PO4−, HPO42−, or PO43−), the feasibility of the sensing platform for ALP assay was validated by evaluating the color changes of ZZ/TiNTs in the presence of PPi, Pi(PO43−), and hydrolysis product of PPi and ALP. As shown in Fig. 4D, ZZ/TiNTs incubated with PPi exhibited a high ΔIred of 72.4, which is much higher than that obtained with a mixture of ALP and PPi or only Pi. In the presence of PPi, the ZC-Zn(Ⅱ) chelate was cleaved by the competitive coordination of PPi-Zn(Ⅱ) framework, and thus unpaired ZC remained on the surface of TiNTs, exhibiting a red color. Compared with PPi, Pi showed a quite low affinity towards Zn2+ ions, and thus no distinct color change can be observed from ZZ/TiNTs. With an incubation time of 60 min (Fig. S6 in Supporting information), PPi can be hydrolyzed by ALP to Pi thoroughly. Based on this hydrolysis, a mixture of ALP and PPi was incubated at 37 ℃ for 60 min, followed by immersing ZZ/TiNTs in the resulting solution. As a result, a negligible color change can be observed from ZZ/TiNTs, indicating the maintenance of ZC-Zn(Ⅱ) in TiNTs.
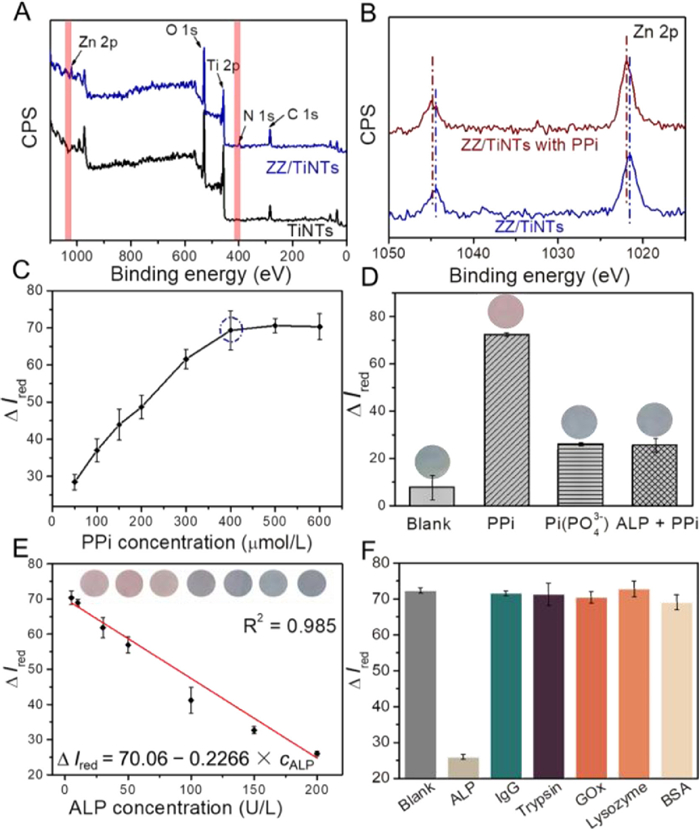
The analytical performance of the proposed sensing platform was then evaluated using ALP solution at a clinically relevant concentration range. Prior to detection, the colorimetric assay was evaluated by varying the reaction time from 5 min to 30 min (Fig. S7A in Supporting information), and pH from 2.5 to 11.5 (Fig. S7B in Supporting information). The optimal reaction time and pH are 15 min and 8.5, respectively. Based on the calibration curve plotted in Fig. 4E, the ΔIred of ZZ/TiNTs displayed a good linear profile from 2 U/L to 200 U/L, with the square of linear correlation coefficient (R2) of 0.985. The limit of detection (LOD) was estimated to be 1 U/L (3σ/s, where σ is the standard deviation of the blank, and s is the slope of the calibration plot). Furthermore, the selectivity of the proposed sensing platform towards ALP is shown in Fig. 4F in the presence of several potential interferences including immunoglobulin G (IgG), trypsin, glucose oxidase (GOx), lysozyme, and bovine serum albumin (BSA). Compared with the high ΔIred value of ZZ/TiNTs loaded with only PPi (blank), the presence of ALP can lead to a clear decrease of ΔIred value, while the presence of other interfering proteins would not trigger the decrease of ΔIred value. These interfering proteins did not influence the decomposition of ZC-Zn(Ⅱ) stimulated by PPi, consistent with the specific hydrolytic ability of ALP towards PPi. The reusability of the proposed sensing platform was investigated by refreshing in a Zn(Ⅱ) containing solution after ALP detection. The Ired values for ALP sensing and after the refreshed treatment were recorded in Fig. S8A (Supporting information). In five cycles, the same sample showed satisfactory repeatability. Moreover, the sensing platform showed a good stability after storage in air for 10 days (Fig. S8B in Supporting information).
A practical application of the sensing platform was performed using diluted human serum samples with the addition of standard concentrations of ALP. The prepared spiked serum samples with additional ALP and PPi were first incubated at 37 ℃ for 60 min, and then ZZ/TiNTs were dipped in this solution for another 15 min to measure the color changes. The results in Table S1 (Supporting information) showed that the recoveries of ALP in diluted serum samples are in the range of 103.5%−107.7%. Compared to the commercial ALP ELISA kit, no significant difference was observed. Therefore, this sensing system was found to be suitable for the qualitative analysis of ALP in a complex biological sample with good recovery and precision.
In conclusion, we have successfully developed a naked-eye colorimetric sensing platform based on a freestanding TiNTs membrane. The high aspect ratio and perpendicular TiO2 nano test tubes in TiNTs provided a long observation length for colorimetric assay. By combining the photocatalysis property of TiO2 and surface functionalization strategy, hydrophilic compartments with a minimal volume were designed on TiNTs as nanoreactors, which can limit indicators and analyst in a confined void, thus improving the sensitivity. ALP concentrations ranging from 2 U/L to 200 U/L can be detected by the proposed colorimetric sensing platform with a high selectivity against other proteins and biological species. The proposed TiNT-based sensing platform paves a new way for constructing a miniaturized and easily implementable POCT device for the monitoring of routine biomarkers.
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
We acknowledge the financial support from the National Natural Science Foundation of China (Nos. 21874013, 22204016 and 22074013), and the Fundamental Research Funds for the Central Universities (Nos. N2005027, N2105018, and N2205005).
Supplementary material associated with this article can be found, in the online version, at doi:
E. Amata, N.D. Bland, R.K. Campbell, M.P. Pollastri, Tetrahedron Lett. 56 (2015) 2832–2835.
doi: 10.1016/j.tetlet.2015.04.061
H. Zou, Z.L. Wang, Y. Cao, G. Huang, Chin. Chem. Lett. 29 (2018) 1355–1358.
doi: 10.1016/j.cclet.2017.10.034
W. Luo, B. Shao, J. Li, et al., Org. Chem. Front. 7 (2020) 1016–1021.
doi: 10.1039/d0qo00140f
S. Zhang, G. Xu, H. Yan, et al., Chin. Chem. Lett. 33 (2022) 5128–5131.
doi: 10.1016/j.cclet.2022.04.006
X. Han, W. Yao, T. Wang, et al., Angew. Chem. Int. Ed. 53 (2014) 5643–5647.
doi: 10.1002/anie.201311214
D. Hack, A.B. Dürr, K. Deckers, et al., Angew. Chem. Int. Ed. 55 (2016) 1797–1800.
doi: 10.1002/anie.201510602
L. Wang, S. Li, P. Chauhan, et al., Chem. Eur. J. 22 (2016) 5123–5127.
doi: 10.1002/chem.201600515
J. Zheng, S.B. Wang, C. Zheng, S.L. You, Angew. Chem. Int. Ed. 56 (2017) 4540–4544.
doi: 10.1002/anie.201700021
J.R. Chen, X.Q. Hu, L.Q. Lu, W.J. Xiao, Chem. Rev. 115 (2015) 5301–5365.
doi: 10.1021/cr5006974
C. Zhu, Y. Ding, L.W. Ye, Org. Biomol. Chem. 13 (2015) 2530–2536.
doi: 10.1039/C4OB02556C
B. Xiang, Y. Wang, C. Xiao, F. He, Y. Huang, Chin. Chem. Lett. 35 (2024) 108777.
doi: 10.1016/j.cclet.2023.108777
D.T. Ziegler, L. Riesgo, T. Ikeda, Y. Fujiwara, G.C. Fu, Angew. Chem. Int. Ed. 53 (2014) 13183–13187.
doi: 10.1002/anie.201405854
Q. Zhang, L. Yang, X. Tong, J. Am. Chem. Soc. 132 (2010) 2550–2551.
doi: 10.1021/ja100432m
S. Kramer, G.C. Fu, J. Am. Chem. Soc. 137 (2015) 3803–3806.
doi: 10.1021/jacs.5b01944
L. Li, P. Luo, Y. Deng, Z. Shao, Angew. Chem. Int. Ed. 58 (2019) 4710–4713.
doi: 10.1002/anie.201901511
X.N. Zhang, H.P. Deng, L. Huang, Y. Wei, M. Shi, Chem. Commun. 48 (2012) 8664–8666.
doi: 10.1039/c2cc34619b
X.N. Zhang, G.Q. Chen, X.Y. Tang, Y. Wei, M. Shi, Angew. Chem. Int. Ed. 53 (2014) 10768–10773.
doi: 10.1002/anie.201406100
Y.Y. Zhou, C. Uyeda, Science 363 (2019) 857–862.
doi: 10.1126/science.aau0364
M.J. Behlen, C. Uyeda, J. Am. Chem. Soc. 142 (2020) 17294–17300.
doi: 10.1021/jacs.0c08262
Z. Zhang, F. Xiao, H.M. Wu, X.Q. Dong, C.J. Wang, Org. Lett. 22 (2020) 569–574.
doi: 10.1021/acs.orglett.9b04341
Q.Y. Liao, C. Ma, Y.C. Wang, et al., Chin. Chem. Lett. 34 (2023) 108371.
doi: 10.1016/j.cclet.2023.108371
L. Zhang, J. Zhang, J. Ma, D.J. Cheng, B. Tan, J. Am. Chem. Soc. 139 (2017) 1714–1717.
doi: 10.1021/jacs.6b09634
K.W. Chen, Z.H. Chen, S. Yang, et al., Angew. Chem. Int. Ed. 61 (2022) e202116829.
doi: 10.1002/anie.202116829
Y. Gao, L.Y. Wang, T. Zhang, B.M. Yang, Y. Zhao, Angew. Chem. Int. Ed. 61 (2022) e202200371.
doi: 10.1002/anie.202200371
S. Matsumura, Y. Maeda, T. Nishimura, S. Uemura, J. Am. Chem. Soc. 125 (2003) 8862–8869.
doi: 10.1021/ja035293l
M. Shigeno, T. Yamamoto, M. Murakami, Chem. Eur. J. 15 (2009) 12929–12931.
doi: 10.1002/chem.200902593
N. Ishida, S. Sawano, Y. Masuda, M. Murakami, J. Am. Chem. Soc. 134 (2012) 17502–17504.
doi: 10.1021/ja309013a
N. Ishida, S. Sawano, M. Murakami, Nat. Commun. 5 (2014) 3111.
doi: 10.1038/ncomms4111
Y. Xia, Z. Liu, Z. Liu, et al., J. Am. Chem. Soc. 136 (2014) 3013–3015.
doi: 10.1021/ja500118w
A. Yada, S. Fujita, M. Murakami, J. Am. Chem. Soc. 136 (2014) 7217–7220.
doi: 10.1021/ja502229c
C. Zhang, X.H. Hu, Y.H. Wang, et al., J. Am. Chem. Soc. 134 (2012) 9585–9588.
doi: 10.1021/ja303129s
L. Shao, Y.H. Wang, D.Y. Zhang, J. Xu, X.P. Hu, Angew. Chem. Int. Ed. 55 (2016) 5014–5018.
doi: 10.1002/anie.201510793
X. Gao, R. Cheng, Y.L. Xiao, X.L. Wan, X. Zhang, Chem 5 (2019) 2987–2999.
doi: 10.1016/j.chempr.2019.09.012
R.Z. Li, D.Q. Liu, D. Niu, Nat. Catal. 3 (2020) 672–680.
doi: 10.1038/s41929-020-0462-9
L. Peng, Z. He, X. Xu, C. Guo, Angew. Chem. Int. Ed. 59 (2020) 14270–14274.
doi: 10.1002/anie.202005019
S.J. Li, J. Huang, J.Y. He, et al., RSC Adv. 10 (2020) 38478–38483.
doi: 10.1039/d0ra07698h
J. Huang, H.H. Kong, S.J. Li, et al., Chem. Commun. 57 (2021) 4674–4677.
doi: 10.1039/d1cc00663k
J.T. Xia, L. Li, X.P. Hu, ACS Catal. 11 (2021) 11843–11848.
doi: 10.1021/acscatal.1c03421
W. Guo, L. Zuo, M. Cui, B. Yan, S. Ni, J. Am. Chem. Soc. 143 (2021) 7629–7634.
doi: 10.1021/jacs.1c03182
Z. Li, D. Li, H. Xiang, et al., Chin. Chem. Lett. 33 (2022) 867–870.
doi: 10.1016/j.cclet.2021.08.009
X. Pu, Q.D. Dang, L. Yang, X. Zhang, D. Niu, Nat. Commun. 13 (2022) 2457.
doi: 10.1038/s41467-022-29986-y
H.D. Qian, Z.H. Li, S. Deng, et al., J. Am. Chem. Soc. 144 (2022) 15779–15785.
doi: 10.1021/jacs.2c06560
R.Q. Wang, C. Shen, X. Cheng, X.Q. Dong, C.J. Wang, Chem. Commun. 58 (2022) 8552–8555.
doi: 10.1039/d2cc01695h
T. Liu, S. Ni, W. Guo, Chem. Sci. 13 (2022) 6806–6812.
doi: 10.1039/d2sc02318k
W. Dong, Z. Zhao, C.Z. Gu, et al., J. Am. Chem. Soc. 145 (2023) 27539–27554.
doi: 10.1021/jacs.3c09155
G. Tian, M. Ji, F. Wu, et al., Org. Lett. 25 (2023) 4666–4671.
doi: 10.1021/acs.orglett.3c01521
C. Gui, Y. Peng, Y. Zhou, et al., ACS Catal. 13 (2023) 13735–13742.
doi: 10.1021/acscatal.3c03814
T. Wang, Y. You, Z.H. Wang, et al., Org. Lett. 25 (2023) 1274–1279.
doi: 10.1021/acs.orglett.3c00075
Z.Q. Geng, C. Zhao, H.D. Qian, et al., Org. Lett. 25 (2023) 4504–4509.
doi: 10.1021/acs.orglett.3c01524
Q. Cai, H. Rao, S.J. Li, et al., Chem 10 (2024) 265–282.
doi: 10.1016/j.chempr.2023.09.006
D.Y. Zhang, L. Shao, J. Xu, X.P. Hu, ACS Catal. 5 (2015) 5026–5030.
doi: 10.1021/acscatal.5b01283
T.R. Li, B.Y. Cheng, Y.N. Wang, et al., Angew. Chem. Int. Ed. 55 (2016) 12422–12426.
doi: 10.1002/anie.201605900
X. Lu, L. Ge, C. Cheng, et al., Chem. Eur. J. 23 (2017) 7689–7693.
doi: 10.1002/chem.201701741
H. Chen, X. Lu, X. Xia, et al., Org. Lett. 20 (2018) 1760–1763.
doi: 10.1021/acs.orglett.8b00253
Z.J. Zhang, L. Zhang, R.L. Geng, et al., Angew. Chem. Int. Ed. 58 (2019) 12190–12194.
doi: 10.1002/anie.201907188
B.C. Wang, T. Fan, F.Y. Xiong, et al., J. Am. Chem. Soc. 144 (2022) 19932–19941.
doi: 10.1021/jacs.2c08090
A. Jonek, S. Berger, E. Haak, Chem. Eur. J. 18 (2012) 15504–15511.
doi: 10.1002/chem.201202414
N. Thies, M. Gerlach, E. Haak, Eur. J. Org. Chem. 2013 (2013) 7354–7365.
doi: 10.1002/ejoc.201300803
N. Thies, E. Haak, Angew. Chem. Int. Ed. 54 (2015) 4097–4101.
doi: 10.1002/anie.201412207
S. Niu, Y. Luo, C. Xu, et al., ACS Catal. 12 (2022) 6840–6850.
doi: 10.1021/acscatal.2c00911
H.H. Kong, C. Zhu, S. Deng, et al., J. Am. Chem. Soc. 144 (2022) 21347–21355.
doi: 10.1021/jacs.2c09572
M.D. Li, Z.H. Wang, H. Zhu, et al., Angew. Chem. Int. Ed. 62 (2023) e202313911.
doi: 10.1002/anie.202313911
S.Y. Luo, G.Q. Lin; , Z.T. He, Org. Chem. Front. 11 (2024) 690–695.
doi: 10.1039/d3qo01749d
H.Y. Lu, Y.W. Chen, W.C. Zhao, et al., Nat. Synth. 2 (2023) 37–48.
C. Xu, Y. Luo, S. Niu, et al., Green Synth. Catal. 5 (2024) 303–309.
doi: 10.1016/j.gresc.2023.10.003
H. Zhu, L. Xu, B. Zhu, et al., Org. Lett. 25 (2023) 9213–9218.
doi: 10.1021/acs.orglett.3c03871
Y.Z. Sun, Z.Y. Ren, Y.X. Yang, et al., Angew. Chem. Int. Ed. 62 (2023) e202314517.
doi: 10.1002/anie.202314517
D. Luo, S. Niu, F. Gong, et al., ACS Catal. 14 (2024) 2746–2757.
doi: 10.1021/acscatal.3c06146
H.D. Qian, X. Li, T. Yin, et al., Sci. China Chem. 67 (2024) 1175–1180.
doi: 10.1007/s11426-023-1922-5
J. Song, Z.J. Zhang, L.Z. Gong, Angew. Chem. Int. Ed. 56 (2017) 5212–5216.
doi: 10.1002/anie.201700105
J. Ren, C. Pi, Y. Wu, X. Cui, Org. Lett. 21 (2019) 4067–4071.
doi: 10.1021/acs.orglett.9b01246
Z.L. Li, G.C. Fang, Q.S. Gu, X.Y. Liu, Chem. Soc. Rev. 49 (2020) 32–48.
doi: 10.1039/c9cs00681h
X. Chang, Y. Yang, C. Shen, et al., J. Am. Chem. Soc. 143 (2021) 3519–3535.
doi: 10.1021/jacs.0c12911
Y.H. Wen, Z.J. Zhang, S. Li, J. Song, L.Z. Gong, Nat. Commun. 13 (2022) 1344.
doi: 10.1038/s41467-022-29059-0
Z. Zhang, P. Chen, G. Liu, Chem. Soc. Rev. 51 (2022) 1640–1658.
doi: 10.1039/d1cs00727k
Z. Yu, Z. Li, C. Yang, Q. Gu, X. Liu, Acta Chim. Sin. 81 (2023) 955–966.
doi: 10.6023/a23040161
E. Amata, N.D. Bland, R.K. Campbell, M.P. Pollastri, Tetrahedron Lett. 56 (2015) 2832–2835.
doi: 10.1016/j.tetlet.2015.04.061
H. Zou, Z.L. Wang, Y. Cao, G. Huang, Chin. Chem. Lett. 29 (2018) 1355–1358.
doi: 10.1016/j.cclet.2017.10.034
W. Luo, B. Shao, J. Li, et al., Org. Chem. Front. 7 (2020) 1016–1021.
doi: 10.1039/d0qo00140f
S. Zhang, G. Xu, H. Yan, et al., Chin. Chem. Lett. 33 (2022) 5128–5131.
doi: 10.1016/j.cclet.2022.04.006
X. Han, W. Yao, T. Wang, et al., Angew. Chem. Int. Ed. 53 (2014) 5643–5647.
doi: 10.1002/anie.201311214
D. Hack, A.B. Dürr, K. Deckers, et al., Angew. Chem. Int. Ed. 55 (2016) 1797–1800.
doi: 10.1002/anie.201510602
L. Wang, S. Li, P. Chauhan, et al., Chem. Eur. J. 22 (2016) 5123–5127.
doi: 10.1002/chem.201600515
J. Zheng, S.B. Wang, C. Zheng, S.L. You, Angew. Chem. Int. Ed. 56 (2017) 4540–4544.
doi: 10.1002/anie.201700021
J.R. Chen, X.Q. Hu, L.Q. Lu, W.J. Xiao, Chem. Rev. 115 (2015) 5301–5365.
doi: 10.1021/cr5006974
C. Zhu, Y. Ding, L.W. Ye, Org. Biomol. Chem. 13 (2015) 2530–2536.
doi: 10.1039/C4OB02556C
B. Xiang, Y. Wang, C. Xiao, F. He, Y. Huang, Chin. Chem. Lett. 35 (2024) 108777.
doi: 10.1016/j.cclet.2023.108777
D.T. Ziegler, L. Riesgo, T. Ikeda, Y. Fujiwara, G.C. Fu, Angew. Chem. Int. Ed. 53 (2014) 13183–13187.
doi: 10.1002/anie.201405854
Q. Zhang, L. Yang, X. Tong, J. Am. Chem. Soc. 132 (2010) 2550–2551.
doi: 10.1021/ja100432m
S. Kramer, G.C. Fu, J. Am. Chem. Soc. 137 (2015) 3803–3806.
doi: 10.1021/jacs.5b01944
L. Li, P. Luo, Y. Deng, Z. Shao, Angew. Chem. Int. Ed. 58 (2019) 4710–4713.
doi: 10.1002/anie.201901511
X.N. Zhang, H.P. Deng, L. Huang, Y. Wei, M. Shi, Chem. Commun. 48 (2012) 8664–8666.
doi: 10.1039/c2cc34619b
X.N. Zhang, G.Q. Chen, X.Y. Tang, Y. Wei, M. Shi, Angew. Chem. Int. Ed. 53 (2014) 10768–10773.
doi: 10.1002/anie.201406100
Y.Y. Zhou, C. Uyeda, Science 363 (2019) 857–862.
doi: 10.1126/science.aau0364
M.J. Behlen, C. Uyeda, J. Am. Chem. Soc. 142 (2020) 17294–17300.
doi: 10.1021/jacs.0c08262
Z. Zhang, F. Xiao, H.M. Wu, X.Q. Dong, C.J. Wang, Org. Lett. 22 (2020) 569–574.
doi: 10.1021/acs.orglett.9b04341
Q.Y. Liao, C. Ma, Y.C. Wang, et al., Chin. Chem. Lett. 34 (2023) 108371.
doi: 10.1016/j.cclet.2023.108371
L. Zhang, J. Zhang, J. Ma, D.J. Cheng, B. Tan, J. Am. Chem. Soc. 139 (2017) 1714–1717.
doi: 10.1021/jacs.6b09634
K.W. Chen, Z.H. Chen, S. Yang, et al., Angew. Chem. Int. Ed. 61 (2022) e202116829.
doi: 10.1002/anie.202116829
Y. Gao, L.Y. Wang, T. Zhang, B.M. Yang, Y. Zhao, Angew. Chem. Int. Ed. 61 (2022) e202200371.
doi: 10.1002/anie.202200371
S. Matsumura, Y. Maeda, T. Nishimura, S. Uemura, J. Am. Chem. Soc. 125 (2003) 8862–8869.
doi: 10.1021/ja035293l
M. Shigeno, T. Yamamoto, M. Murakami, Chem. Eur. J. 15 (2009) 12929–12931.
doi: 10.1002/chem.200902593
N. Ishida, S. Sawano, Y. Masuda, M. Murakami, J. Am. Chem. Soc. 134 (2012) 17502–17504.
doi: 10.1021/ja309013a
N. Ishida, S. Sawano, M. Murakami, Nat. Commun. 5 (2014) 3111.
doi: 10.1038/ncomms4111
Y. Xia, Z. Liu, Z. Liu, et al., J. Am. Chem. Soc. 136 (2014) 3013–3015.
doi: 10.1021/ja500118w
A. Yada, S. Fujita, M. Murakami, J. Am. Chem. Soc. 136 (2014) 7217–7220.
doi: 10.1021/ja502229c
C. Zhang, X.H. Hu, Y.H. Wang, et al., J. Am. Chem. Soc. 134 (2012) 9585–9588.
doi: 10.1021/ja303129s
L. Shao, Y.H. Wang, D.Y. Zhang, J. Xu, X.P. Hu, Angew. Chem. Int. Ed. 55 (2016) 5014–5018.
doi: 10.1002/anie.201510793
X. Gao, R. Cheng, Y.L. Xiao, X.L. Wan, X. Zhang, Chem 5 (2019) 2987–2999.
doi: 10.1016/j.chempr.2019.09.012
R.Z. Li, D.Q. Liu, D. Niu, Nat. Catal. 3 (2020) 672–680.
doi: 10.1038/s41929-020-0462-9
L. Peng, Z. He, X. Xu, C. Guo, Angew. Chem. Int. Ed. 59 (2020) 14270–14274.
doi: 10.1002/anie.202005019
S.J. Li, J. Huang, J.Y. He, et al., RSC Adv. 10 (2020) 38478–38483.
doi: 10.1039/d0ra07698h
J. Huang, H.H. Kong, S.J. Li, et al., Chem. Commun. 57 (2021) 4674–4677.
doi: 10.1039/d1cc00663k
J.T. Xia, L. Li, X.P. Hu, ACS Catal. 11 (2021) 11843–11848.
doi: 10.1021/acscatal.1c03421
W. Guo, L. Zuo, M. Cui, B. Yan, S. Ni, J. Am. Chem. Soc. 143 (2021) 7629–7634.
doi: 10.1021/jacs.1c03182
Z. Li, D. Li, H. Xiang, et al., Chin. Chem. Lett. 33 (2022) 867–870.
doi: 10.1016/j.cclet.2021.08.009
X. Pu, Q.D. Dang, L. Yang, X. Zhang, D. Niu, Nat. Commun. 13 (2022) 2457.
doi: 10.1038/s41467-022-29986-y
H.D. Qian, Z.H. Li, S. Deng, et al., J. Am. Chem. Soc. 144 (2022) 15779–15785.
doi: 10.1021/jacs.2c06560
R.Q. Wang, C. Shen, X. Cheng, X.Q. Dong, C.J. Wang, Chem. Commun. 58 (2022) 8552–8555.
doi: 10.1039/d2cc01695h
T. Liu, S. Ni, W. Guo, Chem. Sci. 13 (2022) 6806–6812.
doi: 10.1039/d2sc02318k
W. Dong, Z. Zhao, C.Z. Gu, et al., J. Am. Chem. Soc. 145 (2023) 27539–27554.
doi: 10.1021/jacs.3c09155
G. Tian, M. Ji, F. Wu, et al., Org. Lett. 25 (2023) 4666–4671.
doi: 10.1021/acs.orglett.3c01521
C. Gui, Y. Peng, Y. Zhou, et al., ACS Catal. 13 (2023) 13735–13742.
doi: 10.1021/acscatal.3c03814
T. Wang, Y. You, Z.H. Wang, et al., Org. Lett. 25 (2023) 1274–1279.
doi: 10.1021/acs.orglett.3c00075
Z.Q. Geng, C. Zhao, H.D. Qian, et al., Org. Lett. 25 (2023) 4504–4509.
doi: 10.1021/acs.orglett.3c01524
Q. Cai, H. Rao, S.J. Li, et al., Chem 10 (2024) 265–282.
doi: 10.1016/j.chempr.2023.09.006
D.Y. Zhang, L. Shao, J. Xu, X.P. Hu, ACS Catal. 5 (2015) 5026–5030.
doi: 10.1021/acscatal.5b01283
T.R. Li, B.Y. Cheng, Y.N. Wang, et al., Angew. Chem. Int. Ed. 55 (2016) 12422–12426.
doi: 10.1002/anie.201605900
X. Lu, L. Ge, C. Cheng, et al., Chem. Eur. J. 23 (2017) 7689–7693.
doi: 10.1002/chem.201701741
H. Chen, X. Lu, X. Xia, et al., Org. Lett. 20 (2018) 1760–1763.
doi: 10.1021/acs.orglett.8b00253
Z.J. Zhang, L. Zhang, R.L. Geng, et al., Angew. Chem. Int. Ed. 58 (2019) 12190–12194.
doi: 10.1002/anie.201907188
B.C. Wang, T. Fan, F.Y. Xiong, et al., J. Am. Chem. Soc. 144 (2022) 19932–19941.
doi: 10.1021/jacs.2c08090
A. Jonek, S. Berger, E. Haak, Chem. Eur. J. 18 (2012) 15504–15511.
doi: 10.1002/chem.201202414
N. Thies, M. Gerlach, E. Haak, Eur. J. Org. Chem. 2013 (2013) 7354–7365.
doi: 10.1002/ejoc.201300803
N. Thies, E. Haak, Angew. Chem. Int. Ed. 54 (2015) 4097–4101.
doi: 10.1002/anie.201412207
S. Niu, Y. Luo, C. Xu, et al., ACS Catal. 12 (2022) 6840–6850.
doi: 10.1021/acscatal.2c00911
H.H. Kong, C. Zhu, S. Deng, et al., J. Am. Chem. Soc. 144 (2022) 21347–21355.
doi: 10.1021/jacs.2c09572
M.D. Li, Z.H. Wang, H. Zhu, et al., Angew. Chem. Int. Ed. 62 (2023) e202313911.
doi: 10.1002/anie.202313911
S.Y. Luo, G.Q. Lin; , Z.T. He, Org. Chem. Front. 11 (2024) 690–695.
doi: 10.1039/d3qo01749d
H.Y. Lu, Y.W. Chen, W.C. Zhao, et al., Nat. Synth. 2 (2023) 37–48.
C. Xu, Y. Luo, S. Niu, et al., Green Synth. Catal. 5 (2024) 303–309.
doi: 10.1016/j.gresc.2023.10.003
H. Zhu, L. Xu, B. Zhu, et al., Org. Lett. 25 (2023) 9213–9218.
doi: 10.1021/acs.orglett.3c03871
Y.Z. Sun, Z.Y. Ren, Y.X. Yang, et al., Angew. Chem. Int. Ed. 62 (2023) e202314517.
doi: 10.1002/anie.202314517
D. Luo, S. Niu, F. Gong, et al., ACS Catal. 14 (2024) 2746–2757.
doi: 10.1021/acscatal.3c06146
H.D. Qian, X. Li, T. Yin, et al., Sci. China Chem. 67 (2024) 1175–1180.
doi: 10.1007/s11426-023-1922-5
J. Song, Z.J. Zhang, L.Z. Gong, Angew. Chem. Int. Ed. 56 (2017) 5212–5216.
doi: 10.1002/anie.201700105
J. Ren, C. Pi, Y. Wu, X. Cui, Org. Lett. 21 (2019) 4067–4071.
doi: 10.1021/acs.orglett.9b01246
Z.L. Li, G.C. Fang, Q.S. Gu, X.Y. Liu, Chem. Soc. Rev. 49 (2020) 32–48.
doi: 10.1039/c9cs00681h
X. Chang, Y. Yang, C. Shen, et al., J. Am. Chem. Soc. 143 (2021) 3519–3535.
doi: 10.1021/jacs.0c12911
Y.H. Wen, Z.J. Zhang, S. Li, J. Song, L.Z. Gong, Nat. Commun. 13 (2022) 1344.
doi: 10.1038/s41467-022-29059-0
Z. Zhang, P. Chen, G. Liu, Chem. Soc. Rev. 51 (2022) 1640–1658.
doi: 10.1039/d1cs00727k
Z. Yu, Z. Li, C. Yang, Q. Gu, X. Liu, Acta Chim. Sin. 81 (2023) 955–966.
doi: 10.6023/a23040161

Yan-Bo Li , Yi Li , Liang Yin . Copper(Ⅰ)-catalyzed diastereodivergent construction of vicinal P-chiral and C-chiral centers facilitated by dual "soft-soft" interaction. Chinese Chemical Letters, 2024, 35(7): 109294-. doi: 10.1016/j.cclet.2023.109294
Zhirong Yang , Shan Wang , Ming Jiang , Gengchen Li , Long Li , Fangzhi Peng , Zhihui Shao . One stone three birds: Ni-catalyzed asymmetric allenylic substitution of allenic ethers, hydroalkylation of 1,3-enynes and double alkylation of enynyl ethers. Chinese Chemical Letters, 2024, 35(8): 109518-. doi: 10.1016/j.cclet.2024.109518
Jing-Qi Tao , Shuai Liu , Tian-Yu Zhang , Hong Xin , Xu Yang , Xin-Hua Duan , Li-Na Guo . Photoinduced copper-catalyzed alkoxyl radical-triggered ring-expansion/aminocarbonylation cascade. Chinese Chemical Letters, 2024, 35(6): 109263-. doi: 10.1016/j.cclet.2023.109263
Yanxin Jiang , Kwai Wun Cheng , Zhiping Yang , Jun (Joelle) Wang . Pd-catalyzed enantioselective and regioselective asymmetric hydrophosphorylation and hydrophosphinylation of enynes. Chinese Chemical Letters, 2025, 36(5): 110231-. doi: 10.1016/j.cclet.2024.110231
Xiaohui Fu , Yanping Zhang , Juan Liao , Zhen-Hua Wang , Yong You , Jian-Qiang Zhao , Mingqiang Zhou , Wei-Cheng Yuan . Palladium-catalyzed enantioselective decarboxylation of vinyl cyclic carbamates: Generation of amide-based aza-1,3-dipoles and application to asymmetric 1,3-dipolar cycloaddition. Chinese Chemical Letters, 2024, 35(12): 109688-. doi: 10.1016/j.cclet.2024.109688
Wujun Jian , Mong-Feng Chiou , Yajun Li , Hongli Bao , Song Yang . Cu-catalyzed regioselective diborylation of 1,3-enynes for the efficient synthesis of 1,4-diborylated allenes. Chinese Chemical Letters, 2024, 35(5): 108980-. doi: 10.1016/j.cclet.2023.108980
Xingfen Huang , Jiefeng Zhu , Chuan He . Catalytic enantioselective N-silylation of sulfoximine. Chinese Chemical Letters, 2024, 35(4): 108783-. doi: 10.1016/j.cclet.2023.108783
Yuemin Chen , Yunqi Wu , Guoao Wang , Feihu Cui , Haitao Tang , Yingming Pan . Electricity-driven enantioselective cross-dehydrogenative coupling of two C(sp3)-H bonds enabled by organocatalysis. Chinese Chemical Letters, 2024, 35(9): 109445-. doi: 10.1016/j.cclet.2023.109445
Chengyao Zhao , Jingyuan Liao , Yuxiang Zhu , Yiying Zhang , Lianjie Zhai , Junrong Huang , Hengzhi You . Polystyrene-supported phosphoric-acid catalyzed atroposelective construction of axially chiral N-aryl benzimidazoles. Chinese Chemical Letters, 2025, 36(6): 110337-. doi: 10.1016/j.cclet.2024.110337
Jieshuai Xiao , Yuan Zheng , Yue Zhao , Zhuangzhi Shi , Minyan Wang . Asymmetric Nozaki-Hiyama-Kishi (NHK)-type reaction of isatins with aromatic iodides by cobalt catalysis. Chinese Chemical Letters, 2025, 36(5): 110243-. doi: 10.1016/j.cclet.2024.110243
Ruilong Geng , Lingzi Peng , Chang Guo . Dynamic kinetic stereodivergent transformations of propargylic ammonium salts via dual nickel and copper catalysis. Chinese Chemical Letters, 2024, 35(8): 109433-. doi: 10.1016/j.cclet.2023.109433
Rong-Nan Yi , Wei-Min He . Visible light/copper catalysis enabled radial type ring-opening of sulfonium salts. Chinese Chemical Letters, 2025, 36(4): 110787-. doi: 10.1016/j.cclet.2024.110787
Conghui Wang , Lei Xu , Zhenhua Jia , Teck-Peng Loh . Recent applications of macrocycles in supramolecular catalysis. Chinese Chemical Letters, 2024, 35(4): 109075-. doi: 10.1016/j.cclet.2023.109075
Wei Chen , Pieter Cnudde . A minireview to ketene chemistry in zeolite catalysis. Chinese Journal of Structural Chemistry, 2024, 43(11): 100412-100412. doi: 10.1016/j.cjsc.2024.100412
Lin Zhang , Chaoran Li , Thongthai Witoon , Xingda An , Le He . Nano-thermometry in photothermal catalysis. Chinese Journal of Structural Chemistry, 2025, 44(4): 100456-100456. doi: 10.1016/j.cjsc.2024.100456
Pengzi Wang , Wenjing Xiao , Jiarong Chen . Copper-Catalyzed C―O Bond Formation by Kharasch-Sosnovsky-Type Reaction. University Chemistry, 2025, 40(4): 239-244. doi: 10.12461/PKU.DXHX202406090
Yu Mao , Yilin Liu , Xiaochen Wang , Shengyang Ni , Yi Pan , Yi Wang . Acylfluorination of enynes via phosphine and silver catalysis. Chinese Chemical Letters, 2024, 35(8): 109443-. doi: 10.1016/j.cclet.2023.109443
Jiaqi Jia , Kathiravan Murugesan , Chen Zhu , Huifeng Yue , Shao-Chi Lee , Magnus Rueping . Multiphoton photoredox catalysis enables selective hydrodefluorinations. Chinese Chemical Letters, 2025, 36(2): 109866-. doi: 10.1016/j.cclet.2024.109866
Tingyu Zhu , Hui Zhang , Wenwei Zhang . Exploration and Practice of Ideological and Political Education in the Course of Experiments on Chemical Functional Molecules: Synthesis and Catalytic Performance Study of Chiral Mn(III)Cl-Salen Complex. University Chemistry, 2024, 39(4): 75-80. doi: 10.3866/PKU.DXHX202311011
Hong Lu , Yidie Zhai , Xingxing Cheng , Yujia Gao , Qing Wei , Hao Wei . Advancements and Expansions in the Proline-Catalyzed Asymmetric Aldol Reaction. University Chemistry, 2024, 39(5): 154-162. doi: 10.3866/PKU.DXHX202310074