An AIEgen nano-assembly for simultaneous detection of ATP and H2S
-
* Corresponding author.
E-mail address: cqyong@ncu.edu.cn (Q.-Y. Cao).
1 These authors contributed equally to this work.
Citation:
Jia-Mei Qin, Xue Li, Wei Lang, Fu-Hao Zhang, Qian-Yong Cao. An AIEgen nano-assembly for simultaneous detection of ATP and H2S[J]. Chinese Chemical Letters,
;2024, 35(6): 108925.
doi:
10.1016/j.cclet.2023.108925

Adenosine triphosphate (ATP), known as "the energy currency of the cell", plays an important role in living cells, including protein synthesis, intracellular signal transduction and cell division [1,2]. The abnormal levels of intracellular ATP have been proved to be closely associated with a variety of diseases, such as cancer and Parkinson [3]. On the other hand, as a signal molecule, hydrogen sulfide (H2S) widely exists in heart, brain, liver and other major organs, and plays an irreplaceable role in physiological functions including neural regulation, apoptosis, insulin signal inhibition and blood pressure [4]. The normal concentration of H2S in cells is about 0.01–3 µmol/L [5]. Abnormal H2S level can lead to the disfunction of cell, which is related to many diseases, like cirrhosis and Alzheimer's disease [6]. Therefore, the detection of ATP and H2S level has important physiological and pathological significance.
Recently, fluorescent sensors of ATP and H2S within cellular environments are well developed for the fast response, excellent selectivity, high sensitivity and simple operation of the fluorescent technique [7]. The ATP sensors are mainly on the basis of the different host-guest interaction mechanism, for instance, metal ion complexation, hydrogen bonding, π-π interaction and the electrostatic interaction [8–12], while the recognition of H2S is based on the different types of chemical reaction, including copper sulfide precipitation, nucleophilic addition and reductive reaction [13,14]. Nevertheless, the traditional sensors are mainly limited to detect single species, fluorescent sensors for the simultaneous recognition of ATP and H2S are still rare.
Currently, fluorescent sensors for simultaneous detection dual or multiple chemical species in biology have attracted much attention [15–17]. Some multifunctional fluorescent sensors for ATP and another analyte, including H2O2, H2S, ONOO− and nitroreductase (NTR), were also documented [18–23]. Typically, the multi-analyte chemosensors were elegantly prepared by covalently linked multiple analyte recognition sites, including complexation and chemical reaction, with one or two flurophores in a single molecule. This strategy requires cumbersome chemical synthesis methods and is time-consuming and labor-intensive.
Aggregation-induced emission (AIE) fluorogens, also named as AIEgens, which have attracted much attention for their unique fluorescence emission properties [24]. The AIEgens show non- or weak emission in dilution solutions, but emit strongly at the aggregation state. Previously, we have reported imidazolium-functionalized tetraphenylethylenes, which have a good fluorescence turn-on sensing toward ATP for the complexation enhanced aggregation [25]. We envisioned that if the second recognition site such as the disulfide bond is rationally incorporated into this system, a dual functional probe which can detect ATP and another analyte will be obtained. In this context, herein, we design and synthesize a novel AIE bifunctional probe TPEPy-SS-C14 (Scheme 1), which can simultaneously detect ATP and H2S based on the aggregation/disaggregation mechanism. The probe TPEPy-SS-C14 is rationally designed as following: (1) The TPEPy unit makes probe a red AIE-based emission, (2) the pyridinium and amide groups are used as the ATP binding site via the electrostatic interactions and hydrogen bonding, (3) the disulfide bond is cleavable by H2S, and (4) the long alkyl chain endows probe a good amphiphilic property in aqueous solution. In addition, the probe locates mitochondria, and can detect ATP and hydrogen sulfide levels in living cells.
The detailed synthesis route of TPEPy-SS-C14 is shown in Scheme S1 (Supporting information). And the chemical structure of TPEPy-SS-C14 was verified by high-resolution mass spectrometry (HRMS), 1H nuclear magnetic resonance (NMR) and 13C NMR spectra (Figs. S1-S5 in Supporting information).
Firstly, the ultraviolet–visible (UV–vis) absorption and fluorescence spectra of TPEPy-SS-C14 (10 µmol/L) was investigated in aqueous solution. As shown in Fig. 1a, TPEPy-SS-C14 exhibits a red emission with the maximum peak at 630 nm and a low-energy absorption peak at 428 nm. The Stokes shift is 202 nm, which can prevent the interference effectively caused by self-absorption in the excitation process of biological imaging.
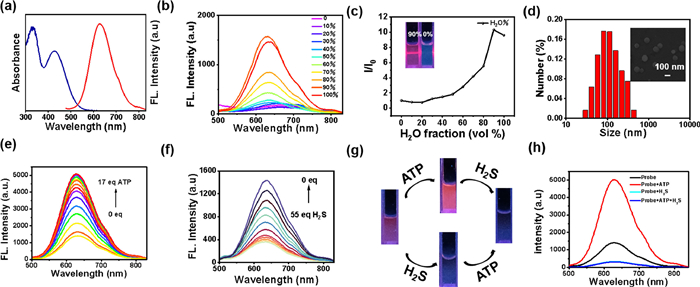
Next, the AIE properties of TPEPy-SS-C14 were studied by emission spectra in water/DMSO mixture (Figs. 1b and c). In DMSO, the soluble TPEPy-SS-C14 molecule shows very weak emission at 630 nm. The fluorescence intensity of TPEPy-SS-C14 increased gradually with the increasing fraction of water and attained the maximum intensity in 90% aqueous solution, which may be due to the formation of nano aggregates. Additionally, the Tyndall experiment, scanning electron microscopy (SEM) and dynamic light scattering (DLS) measurements well confirmed the formation of nona-aggregation. The TPEPy-SS-C14 aggregations show a sphere morphology with a diameter of 160–180 nm (Fig. 1d). Besides, the critical micelle concentration (CMC) of TPEPy-SS-C14 was also investigated by concentration-dependent fluorescence experiment in aqueous solution, with the calculated value of 7.78 µmol/L, lower than the concentration used for testing (10 µmol/L, Fig. S6 in Supporting information).
We evaluated the sensing properties of TPEPy-SS-C14 toward ATP in 4-(2-hydroxyethyl)–1-piperazineethanesulfonic acid (HEPES) aqueous buffer solution (pH 7.4). With the addition of ATP, the fluorescence of TPEPy-SS-C14 gradually increased and got saturated when 17 equiv. ATP was added (Fig. 1e), with the fluorescence emission intensity at 630 nm about 3.6-fold enhancement. In addition, according to fluorescence titration data, the binding constant of TPEPy-SS-C14 and ATP is calculated to be 2.57 × 105 mol/L and the stoichiometric binding ratio is 1:1 (Fig. S7a in Supporting information). The detection limit of TPEPy-SS-C14 towards ATP is 12.3 nmol/L (Fig. S7b in Supporting information), indicating that the probe can recognize ATP quantitatively and effectively in physiological conditions.
Next, the fluorescence responding of TPEPy-SS-C14 toward H2S alone was also investigated. In Fig. 1f, upon the increasing concentration of sodium sulfide, the fluorescence of TPEPy-SS-C14 will gradually decrease, with the quenching ratio about 80% when 55 equiv. sodium sulfide was added. This change can be well observed by the naked eye under the irradiation of a 365 nm ultraviolet lamp, along with the fluorescence color varying from red fluorescence to dark luminescence. The fluorescence intensity at 630 nm versus the concentration of hydrogen sulfide exhibits a great linear relation from 0 to 200 µmol/L, and the calculated limit of detection (LOD) is 5.0 × 10−7 mol/L (Fig. S8 in Supporting information). Then the time-dependent fluorescence spectra of TPEPy-SS-C14 added with 40 equiv. H2S were investigated. The fluorescence of probe decreases rapidly in a few seconds and basically reaches equilibrium in three minutes, which indicates that TPEPy-SS-C14 can be used for rapid detection of H2S (Fig. S9 in Supporting information).
Compared to other reported probes for simultaneous detection of ATP and H2S (Table S1 in Supporting information), TPEPy-SS-C14 has a lower detection limit and shorter detection time [18–23]. These listed probes are all using rhodamine linked 1,8-naphthalimide as fluorescence signal matrix, which can recognize ATP through the spirolactam ring-opening mechanism of rhodamine and recognize H2S by the reduction of -N3 on 1,8-naphthalimide. These probes have complex synthesis process and single recognition mechanism, but here we design and synthesize a novel AIE bifunctional probe TPEPy-SS-C14, which can simultaneously detect ATP and H2S based on the aggregation/disaggregation mechanism. TPEPy-SS-C14 can detect ATP with a fluorescence enhancement response due to complexation enhanced aggregation. And H2S can quench the fluorescence due to the disaggregation of the cleavable disulfide bond. In addition, the probe locates mitochondria, and can detect ATP and hydrogen sulfide levels in living cells.
The specific guest induced aggregation-disaggregation mechanism of TPEPy-SS-C14 to detect ATP and H2S are shown in Scheme 1. Upon the addition of ATP, strong intermolecular interactions will occur between pyridinium and amide donors of TPEPy-SS-C14 and the negative phosphate groups of ATP. This proposed binding model can be confirmed by 1H NMR titration of TPEPy-SS-C14 with ATP in DMSO-d6/D2O (8:2, v/v) solution, therein a large chemical shift was observed in the pyridinium and amide protons (Fig. S10 in Supporting information). After binding with ATP, the nano-aggregation of TPEPy-SS-C14 became larger, with the DLS data changing from 180 nm to 360 nm in aqueous solution, and the transmission electron microscope (TEM) increasing up to 340 nm in solid state (Fig. S11 in Supporting information). Thus, the complexation of ATP can enhance the aggregation of TPEPy-SS-C14, which makes the fluorescence a turn-on response. We also investigated the reaction mechanism of TPEPy-SS-C14 toward H2S by high-resolution mass spectrum. In Fig. S12 (Supporting information), after treated with H2S, TPEPy-SS-C14 is cleaved into TPEPy-SSH, C14-SSH and C14-SS-C14 species, indication that the addition of H2S leads to disaggregation.
Next, we studied the sensing performance of TPEPy-SS-C14 in the simultaneous detection of ATP and H2S in HEPES buffer. TPEPy-SS-C14 still can detect H2S effectively in the presence of ATP, but can not recognize ATP with the coexistence in H2S (Figs. 1g and h). We further tested the concentration dependent change of H2S added with ATP. From Fig. S13 (Supporting information), we can see that the fluorescence of TPEPy-SS-C14 at 630 nm first increase upon addition of ATP for complexation. The fluorescence intensity gradually decreases when further addition of H2S, with the quenching ratio as high as 94%. Interesting, the calculated LOD of TPEPy-SS-C14 /ATP toward H2S is 1.57 × 10−7 mol/L, which is lower than that of TPEPy-SS-C14 (5.0 × 10−7 mol/L). This result is reasonable because the H2S trigged disulfide bond cleavage can happen in both TPEPy-SS-C14 and TPEPy-SS-C14 /ATP nano-aggregation. The addition of ATP will not affect the quenching effect of hydrogen sulfide on TPEPy-SS-C14 probe.
To further study the specificity of TPEPy-SS-C14 for ATP and H2S, the responses of the probe to other interfering species, including metal cations, various anions and amino acids were also studied (Fig. 2a). As shown in Fig. 2b and Fig. S14 (Supporting information), a few other phosphate anions, for example, adenosine diphosphate (ADP), adenosine monophosphate (AMP), pyrophosphate (PPi) and inorganic phosphate (Pi), can also slightly enhance the fluorescence. A weak response to ADP, PPi, Pi and AMP, with a partial enhance of the emission spectra of TPEPy-SS-C14 was recorded in Figs. S15 and S16 (Supporting information). However, this interference can be ignored because TPEPy-SS-C14 exhibits far better binding affinity of ATP than these similar structured polyphostates. The detection limits and binding constants of TPEPy-SS-C14 to these anions were also shown in Table S2 (Supporting information). Particularly, the KATP/KADP ratio is about 230.
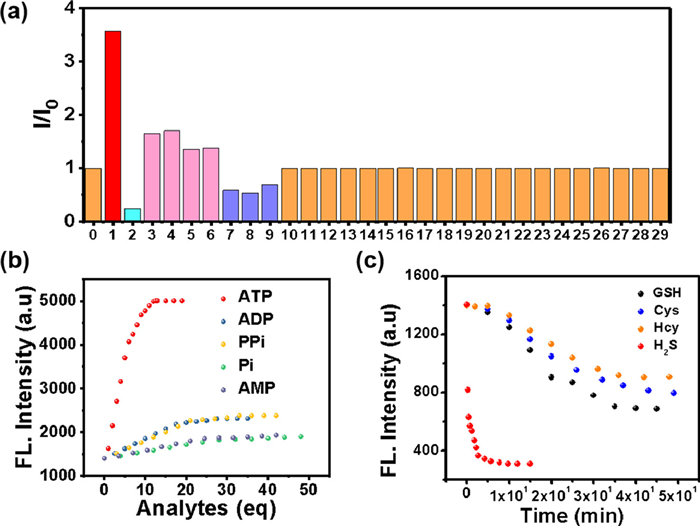
On the other hand, we find the addition of some thiols-containing amide acid, including glutathione (GSH), cysteine (Cys) and homocysteine (Hcy), can also leads to a little decrease of the fluorescence. However, the quench ratio by the thiols is much lower than that of H2S, indicating that TPEPy-SS-C14 can discriminate H2S from thiols and other analytes. The difference fluorescence response of TPEPy-SS-C14 toward H2S and thiols may be attributed their different reaction mechanism. For H2S, which mainly exists as HS− in physiological conditions, can effectively cleave the disulfide bond to release the fluorescent active species TPEPy-SSH [8–12]. However, the thiols reaction may give both the cleavage and the disulfide exchange products. We also studied the reaction mechanism of GSH with the probe (Fig. S17 in Supporting information), which captures not only the cleavage product TPEPy-SH, but also the disulfide exchange species TPEPy-SG. Regarding that TPEPy-SG is also amphiphily for bearing the hydrophilic glutathione group, it may exhibit self-assembly property with high emission. Therefore, the fluorescence is only slightly reduced after adding GSH and other thiols [26]. Furthermore, H2S shows stronger nucleophilicity than bio-thiols. We compared the time kinetics of H2S and other thiols with TPEPy-SS-C14 (Fig. 2c), and found that the fluorescence of hydrogen sulfide added decreased rapidly in a few seconds, and basically reached equilibrium in 5 min, while other thiols took nearly 50 min. So probe TPEPy-SS-C14 reacting with H2S is dominant under the complexed physiological conditions.
For really application, a good multifunctional probe should also behaves an excellent selectively in many competitive ions. Thus, the competitive experiments of TPEPy-SS-C14 toward ATP/H2S under the coexistence of many interference species were carried out (Fig. S18 in Supporting information). It was revealed that TPEPy-SS-C14 still displays a excellent fluorescence enhancement ability toward ATP in the presence of these analytes, except H2S. However, it can recognize H2S in the presence of the following 27 analytes, containing biothiols GSH, Cys and Hcy (Fig. S19 in Supporting information). In addition, the influences of pH experiments reveals that this probe can work in wide pH range of 6‒10, indicating the physiological applicability of the TPEPy-SS-C14 probe (Fig. S20 in Supporting information).
Based on the good selective competition and anti-interference ability of the probe in vitro, TPEPy-SS-C14 was used to recognize ATP and H2S in living cells. Firstly, the toxicity of TPEPy-SS-C14 on SMMC cells was conducted by standard MTT assay. As shown in Fig. S21 (Supporting information), TPEPy-SS-C14 possessed a low cytotoxicity at the test concentration and is appropriate for intracellular imaging. The subcellular location of TPEPy-SS-C14 was investigated by co localization experiment with Mito tracker green. TPEPy-SS-C14 overlaps well with Mito tracker green dye, with an overlap rate of 0.91 (Fig. S22 in Supporting information). These results show that TPEPy-SS-C14 has a good localization effect on mitochondria and provides the imaging ability of ATP and hydrogen sulfide in mitochondria.
In order to investigate the ability of TPEPy-SS-C14 to recognize ATP in cells, we divided the cells into three groups: control group (row a), calcium ion treated (row b) and sodium azide treated (row c) (Fig. 3). Ca2+ increases ATP concentration by activating mitochondrial dehydrogenase and the fluorescence intensity of corresponding cell imaging is 120% of that of the control group. On the contrary, sodium azide will inhibit enzyme activity, leading to the decrease of ATP concentration and the fluorescence intensity of corresponding cell imaging is only 29% of that of the control group. This indicates that the probe can successfully detect the level of ATP in cells.
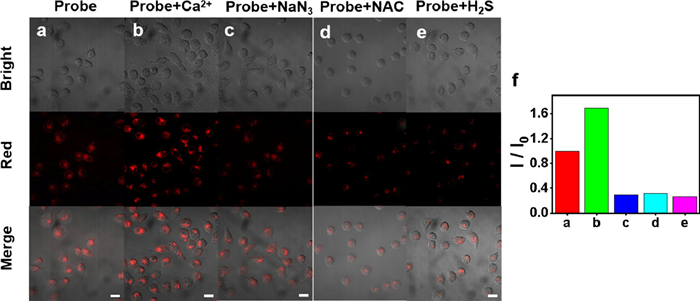
Then, we evaluated the imaging ability of TPEPy-SS-C14 for endogenous and exogenous H2S. In Fig. 3, after incubating the cells pretreated with NAC (1 mmol/L) with 10 µmol/L probes for 1 h, the red fluorescence decreased significantly (row d), indicating that NAC induced the production of endogenous hydrogen sulfide concentration, and leaded to cleavage TPEPy-SS-C14 in living cells. Similarly, the fluorescence of cells treated with exogenous H2S was also reduced obviously as expected (row e). These results demonstrate that TPEPy-SS-C14 can effectively evaluate the level of endogenous and exogenous H2S in cells. Probe TPEPy-SS-C14 can be applied as a tool to explore the level of ATP and hydrogen sulfide in living cell mitochondria.
To sum up, we constructed a dual site AIE fluorescence probe TPEPy-SS-C14 and proposed a new strategy of assembly/disassembly for simultaneous recognition of ATP and H2S. As a proof of concept, the assembly/disassembly mechanism was applied to modulate the fluorescence of the amphiphilic AIEgen. After interaction ATP or H2S with probe, the aggregation state of the probe in the aqueous solution changes, showing that the fluorescence increases or decreases. It is worth mentioning that TPEPy-SS-C14 has good sensitivity and selectivity to ATP and hydrogen sulfide in SMMC cells under physiological conditions. This system has superiority over previously reported work, such as easier synthesis, lower detection limit, shorter detection time and simple operation. The sensor possesses the potential to become a effective tool to research the relationship between ATP and H2S in mitochondria of living cells.
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
This work was supported by the National Nature Science Foundation of China (No. 22061028) and Jiangxi Provincial Natural Science Foundation (No. 20224ACB203012).
Supplementary material associated with this article can be found, in the online version, at doi:
C.F. Higgin, I.D. Hiles, G.P.C. Salmond, et al., Nature 323 (1986) 448–450.
doi: 10.1038/323448a0
P.B. Dennis, A. Jaeschke, M. Saitoh, et al., Science 294 (2001) 1102–1105.
doi: 10.1126/science.1063518
K.T. Bush, S.H. Keller, S.K. Nigam, J. Clin. Invest. 106 (2000) 621–626.
doi: 10.1172/JCI10968
D.H. Matthew, D.P. Michael, Chem Soc. Rev. 45 (2016) 6108–6117.
doi: 10.1039/C6CS00212A
D. Hanahan, A.W. Robert, Cell 144 (2011) 646–674.
doi: 10.1016/j.cell.2011.02.013
D. Giuliani, A. Ottani, D. Zaffe, et al., Neurobiol. Learn. Mem. 104 (2013) 82–91.
doi: 10.1016/j.nlm.2013.05.006
B.H. Huang, B. Liang, R.S. Zhang, D.M. Xing, Coord. Chem. Rev. 452 (2022) 214302.
doi: 10.1016/j.ccr.2021.214302
Y. Zhou, Z.C. Xu, Y.Y. Ju, Chem. Soc. Rev. 40 (2011) 2222–2235.
doi: 10.1039/c0cs00169d
J.B. Stephen, A.J. Katrina, ChemPlusChem 86 (2021) 59–70.
doi: 10.1002/cplu.202000567
X. Zhou, X. Wang, L. Shang, Chin. Chem. Lett. 34 (2023) 108093.
doi: 10.1016/j.cclet.2022.108093
H. Duan, F. Cao, M. Zhang, M. Gao, L. Cao, Chin. Chem. Lett. 33 (2022) 2459–2463.
doi: 10.1016/j.cclet.2021.11.010
L. Jiang, T. Chen, E. Song, Chem. Eng. J. 427 (2022) 131563.
doi: 10.1016/j.cej.2021.131563
M.W. Yang, J.L. Fan, J.J. Du, X.J. Peng, Chem. Sci. 11 (2020) 5127–5141.
doi: 10.1039/d0sc01482f
Z. Fang, Z. Su, W. Qin, et al., Chin. Chem. Lett. 31 (2020) 2903–2908.
doi: 10.1016/j.cclet.2020.03.063
L.L. Wu, J.G. Huang, K.Y. Pu, T.D. James, Nat. Rev. Chem. 5 (2021) 406–421.
doi: 10.1038/s41570-021-00277-2
J.Y. Guo, B. Fang, H. Bai, et al., Trends Anal. Chem. 155 (2022) 116697.
doi: 10.1016/j.trac.2022.116697
J.L. Kolanowski, F. Liu, J. Elizabeth, Chem. Soc. Rev. 47 (2018) 195–208.
doi: 10.1039/C7CS00528H
X. Chai, Z.T. Fan, M.M. Yu, J. Zhao, L.L. Li, Nano Lett. 21 (2021) 10047–10053.
doi: 10.1021/acs.nanolett.1c03732
W.J. Zhang, Y.X. Lu, F.J. Huo, Y.B. Zhang, C.X. Yin, Dyes Pigm. 204 (2022) 110442.
doi: 10.1016/j.dyepig.2022.110442
X.P. Yang, P.Y. Xie, J.F. Liu, et al., Chem. Eng. J. 442 (2022) 136–141.
P.P. Sun, H.C. Chen, S.Y. Lu, et al., Anal. Chem. 94 (2022) 11573–11581.
doi: 10.1021/acs.analchem.2c01850
Y. Fang, W. Shi, Y.M. Hu, X.H. Li, H.M. Ma, Chem. Commun. 54 (2018) 5454–5457.
doi: 10.1039/c8cc02209g
Z. Wu, M.M. Liu, Z.C. Liu, Y. Tian, J. Am. Chem. Soc. 142 (2020) 7532–7541.
doi: 10.1021/jacs.0c00771
L.L. Wu, J.H. Liu, X. Tian, et al., J. Am. Chem. Soc. 144 (2022) 174–183.
doi: 10.1021/jacs.1c07954
H. Tao, L. He, G.J.S. Cheng, Q.Y. Cao, Dyes Pigm. 166 (2019) 233–238.
doi: 10.1016/j.dyepig.2019.03.041
A. Shamirian, H.S. Afsari, D. Wu, L.W. Miller, P.T. Snee, Anal. Chem. 88 (2016) 6050–6056.
doi: 10.1021/acs.analchem.6b01310
C.F. Higgin, I.D. Hiles, G.P.C. Salmond, et al., Nature 323 (1986) 448–450.
doi: 10.1038/323448a0
P.B. Dennis, A. Jaeschke, M. Saitoh, et al., Science 294 (2001) 1102–1105.
doi: 10.1126/science.1063518
K.T. Bush, S.H. Keller, S.K. Nigam, J. Clin. Invest. 106 (2000) 621–626.
doi: 10.1172/JCI10968
D.H. Matthew, D.P. Michael, Chem Soc. Rev. 45 (2016) 6108–6117.
doi: 10.1039/C6CS00212A
D. Hanahan, A.W. Robert, Cell 144 (2011) 646–674.
doi: 10.1016/j.cell.2011.02.013
D. Giuliani, A. Ottani, D. Zaffe, et al., Neurobiol. Learn. Mem. 104 (2013) 82–91.
doi: 10.1016/j.nlm.2013.05.006
B.H. Huang, B. Liang, R.S. Zhang, D.M. Xing, Coord. Chem. Rev. 452 (2022) 214302.
doi: 10.1016/j.ccr.2021.214302
Y. Zhou, Z.C. Xu, Y.Y. Ju, Chem. Soc. Rev. 40 (2011) 2222–2235.
doi: 10.1039/c0cs00169d
J.B. Stephen, A.J. Katrina, ChemPlusChem 86 (2021) 59–70.
doi: 10.1002/cplu.202000567
X. Zhou, X. Wang, L. Shang, Chin. Chem. Lett. 34 (2023) 108093.
doi: 10.1016/j.cclet.2022.108093
H. Duan, F. Cao, M. Zhang, M. Gao, L. Cao, Chin. Chem. Lett. 33 (2022) 2459–2463.
doi: 10.1016/j.cclet.2021.11.010
L. Jiang, T. Chen, E. Song, Chem. Eng. J. 427 (2022) 131563.
doi: 10.1016/j.cej.2021.131563
M.W. Yang, J.L. Fan, J.J. Du, X.J. Peng, Chem. Sci. 11 (2020) 5127–5141.
doi: 10.1039/d0sc01482f
Z. Fang, Z. Su, W. Qin, et al., Chin. Chem. Lett. 31 (2020) 2903–2908.
doi: 10.1016/j.cclet.2020.03.063
L.L. Wu, J.G. Huang, K.Y. Pu, T.D. James, Nat. Rev. Chem. 5 (2021) 406–421.
doi: 10.1038/s41570-021-00277-2
J.Y. Guo, B. Fang, H. Bai, et al., Trends Anal. Chem. 155 (2022) 116697.
doi: 10.1016/j.trac.2022.116697
J.L. Kolanowski, F. Liu, J. Elizabeth, Chem. Soc. Rev. 47 (2018) 195–208.
doi: 10.1039/C7CS00528H
X. Chai, Z.T. Fan, M.M. Yu, J. Zhao, L.L. Li, Nano Lett. 21 (2021) 10047–10053.
doi: 10.1021/acs.nanolett.1c03732
W.J. Zhang, Y.X. Lu, F.J. Huo, Y.B. Zhang, C.X. Yin, Dyes Pigm. 204 (2022) 110442.
doi: 10.1016/j.dyepig.2022.110442
X.P. Yang, P.Y. Xie, J.F. Liu, et al., Chem. Eng. J. 442 (2022) 136–141.
P.P. Sun, H.C. Chen, S.Y. Lu, et al., Anal. Chem. 94 (2022) 11573–11581.
doi: 10.1021/acs.analchem.2c01850
Y. Fang, W. Shi, Y.M. Hu, X.H. Li, H.M. Ma, Chem. Commun. 54 (2018) 5454–5457.
doi: 10.1039/c8cc02209g
Z. Wu, M.M. Liu, Z.C. Liu, Y. Tian, J. Am. Chem. Soc. 142 (2020) 7532–7541.
doi: 10.1021/jacs.0c00771
L.L. Wu, J.H. Liu, X. Tian, et al., J. Am. Chem. Soc. 144 (2022) 174–183.
doi: 10.1021/jacs.1c07954
H. Tao, L. He, G.J.S. Cheng, Q.Y. Cao, Dyes Pigm. 166 (2019) 233–238.
doi: 10.1016/j.dyepig.2019.03.041
A. Shamirian, H.S. Afsari, D. Wu, L.W. Miller, P.T. Snee, Anal. Chem. 88 (2016) 6050–6056.
doi: 10.1021/acs.analchem.6b01310

Jiao Chen , Zihan Zhang , Guojin Sun , Yudi Cheng , Aihua Wu , Zefan Wang , Wenwen Jiang , Fulin Chen , Xiuying Xie , Jianli Li . Benzo[4,5]imidazo[1,2-a]pyrimidine-based structure-inherent targeting fluorescent sensor for imaging lysosomal viscosity and diagnosis of lysosomal storage disorders. Chinese Chemical Letters, 2024, 35(11): 110050-. doi: 10.1016/j.cclet.2024.110050
Qian Ren , Xue Dai , Ran Cen , Yang Luo , Mingyang Li , Ziyun Zhang , Qinghong Bai , Zhu Tao , Xin Xiao . A cucurbit[8]uril-based supramolecular phosphorescent assembly: Cell imaging and sensing of amino acids in aqueous solution. Chinese Chemical Letters, 2024, 35(12): 110022-. doi: 10.1016/j.cclet.2024.110022
Shuangying Li , Qingxiang Zhou , Zhi Li , Menghua Liu , Yanhui Li . Sensitive measurement of silver ions in environmental water samples integrating magnetic ion-imprinted solid phase extraction and carbon dot fluorescent sensor. Chinese Chemical Letters, 2024, 35(5): 108693-. doi: 10.1016/j.cclet.2023.108693
Chuan-Zhi Ni , Ruo-Ming Li , Fang-Qi Zhang , Qu-Ao-Wei Li , Yuan-Yuan Zhu , Jie Zeng , Shuang-Xi Gu . A chiral fluorescent probe for molecular recognition of basic amino acids in solutions and cells. Chinese Chemical Letters, 2024, 35(10): 109862-. doi: 10.1016/j.cclet.2024.109862
Boran Cheng , Lei Cao , Chen Li , Fang-Yi Huo , Qian-Fang Meng , Ganglin Tong , Xuan Wu , Lin-Lin Bu , Lang Rao , Shubin Wang . Fluorine-doped carbon quantum dots with deep-red emission for hypochlorite determination and cancer cell imaging. Chinese Chemical Letters, 2024, 35(6): 108969-. doi: 10.1016/j.cclet.2023.108969
Zhixue Liu , Haiqi Chen , Lijuan Guo , Xinyao Sun , Zhi-Yuan Zhang , Junyi Chen , Ming Dong , Chunju Li . Luminescent terphen[3]arene sulfate-activated FRET assemblies for cell imaging. Chinese Chemical Letters, 2024, 35(9): 109666-. doi: 10.1016/j.cclet.2024.109666
Haibo Wan , Zhengzhong Lv , Jicai Jiang , Xuefeng Cheng , Qingfeng Xu , Haibin Shi , Jianmei Lu . Multidimensional detection of roxarsone via AIE-based sulfates. Chinese Chemical Letters, 2025, 36(3): 110023-. doi: 10.1016/j.cclet.2024.110023
Huakang Zong , Xinyue Li , Yanlin Zhang , Faxun Wang , Xingxing Yu , Guotao Duan , Yuanyuan Luo . Pt/Ti3C2 electrode material used for H2S sensor with low detection limit and high stability. Chinese Chemical Letters, 2025, 36(5): 110195-. doi: 10.1016/j.cclet.2024.110195
Brandon Bishop , Shaofeng Huang , Hongxuan Chen , Haijia Yu , Hai Long , Jingshi Shen , Wei Zhang . Artificial transmembrane channel constructed from shape-persistent covalent organic molecular cages capable of ion and small molecule transport. Chinese Chemical Letters, 2024, 35(11): 109966-. doi: 10.1016/j.cclet.2024.109966
Xu Qu , Pengzhao Wu , Kaixuan Duan , Guangwei Wang , Liang-Liang Gao , Yuan Guo , Jianjian Zhang , Donglei Shi . Self-calibrating probes constructed on a unique dual-emissive fluorescence platform for the precise tracking of cellular senescence. Chinese Chemical Letters, 2024, 35(12): 109681-. doi: 10.1016/j.cclet.2024.109681
Yijian Zhao , Jvzhe Li , Yunyi Shi , Jie Hu , Meiyi Liu , Yao Shen , Xinglin Hou , Qiuyue Wang , Qi Wang , Zhiyi Yao . A label-free and ratiometric fluorescent sensor based on porphyrin-metal-organic frameworks for sensitive detection of ochratoxin A in cereal. Chinese Chemical Letters, 2025, 36(4): 110132-. doi: 10.1016/j.cclet.2024.110132
Junying LI , Xinyan CHEN , Xihui DIAO , Muhammad Yaseen , Chao CHEN , Hao WANG , Chuansong QI , Wei LI . Chiral fluorescent sensor Tb3+@Cd-CP based on camphoric acid for the enantioselective recognition of R- and S-propylene glycol. Chinese Journal of Inorganic Chemistry, 2024, 40(12): 2497-2504. doi: 10.11862/CJIC.20240084
Yudi Cheng , Xiao Wang , Jiao Chen , Zihan Zhang , Jiadong Ou , Mengyao She , Fulin Chen , Jianli Li . A near-infrared fluorescent probe for visualizing transformation pathway of Cys/Hcy and H2S and its applications in living system. Chinese Chemical Letters, 2024, 35(5): 109156-. doi: 10.1016/j.cclet.2023.109156
Lei ZHANG , Cheng HE , Yang JIAO . An azo-based fluorescent probe for the detection of hypoxic tumor cells. Chinese Journal of Inorganic Chemistry, 2025, 41(6): 1162-1172. doi: 10.11862/CJIC.20250081
Lixian Fu , Yiyun Tan , Yue Ding , Weixia Qing , Yong Wang . Water–soluble and polarity–sensitive near–infrared fluorescent probe for long–time specific cancer cell membranes imaging and C. Elegans label. Chinese Chemical Letters, 2024, 35(4): 108886-. doi: 10.1016/j.cclet.2023.108886
Shan Jiang , Lingchen Meng , Wenyue Ma , Qingkai Qi , Wei Zhang , Bin Xu , Leijing Liu , Wenjing Tian . Corrigendum to 'Morphology controllable conjugated network polymers based on AIE-active building block for TNP detection' [Chin. Chem. Lett. 32 (2021) 1037-1040]. Chinese Chemical Letters, 2024, 35(12): 108998-. doi: 10.1016/j.cclet.2023.108998
Linfang ZHANG , Wenzhu YIN , Gui YIN . A 2-dicyanomethylene-3-cyano-4,5,5-trimethyl-2,5-dihydrofuran-based near-infrared fluorescence probe for the detection of hydrogen sulfide and imaging of living cells. Chinese Journal of Inorganic Chemistry, 2025, 41(3): 540-548. doi: 10.11862/CJIC.20240405
Tiancong Shi , Xi Chen , Xiao Zhou , Hongyi Zhang , Fuping Han , Lihan Cai , Wen Sun , Jianjun Du , Jiangli Fan , Xiaojun Peng . Azaindole-based asymmetric pentamethine cyanine dye for mitochondrial pH detection and near-infrared ratiometric fluorescence imaging of mitophagy. Chinese Chemical Letters, 2025, 36(6): 110408-. doi: 10.1016/j.cclet.2024.110408
Wei GAO , Meiqi SONG , Xuan REN , Jianliang BAI , Jing SU , Jianlong MA , Zhijun WANG . A self-calibrating fluorescent probe for the selective detection and bioimaging of HClO. Chinese Journal of Inorganic Chemistry, 2025, 41(6): 1173-1182. doi: 10.11862/CJIC.20250112
Jianqiu Li , Yi Zhang , Songen Liu , Jie Niu , Rong Zhang , Yong Chen , Yu Liu . Cucurbit[8]uril-based non-covalent heterodimer realized NIR cell imaging through topological transformation from nanowire to nanorod. Chinese Chemical Letters, 2024, 35(10): 109645-. doi: 10.1016/j.cclet.2024.109645