Advanced municipal wastewater treatment and simultaneous energy/resource recovery via photo(electro)catalysis
-
* Corresponding author.
E-mail address: zjp_112@126.com (J.-P. Zou).
Citation:
Dengke Wang, Siqi Chen, Shiqin Lai, Weili Dai, Lixia Yang, Lanqing Deng, Mengjuan Suo, Xuyang Wang, Jian-Ping Zou, Sheng-Lian Luo. Advanced municipal wastewater treatment and simultaneous energy/resource recovery via photo(electro)catalysis[J]. Chinese Chemical Letters,
;2023, 34(5): 107861.
doi:
10.1016/j.cclet.2022.107861

Precise tumor targeting and on-demand drug release show great potentials in cancer therapy, which have been achieved by the continuously emerging smart prodrugs activated by the specific stimuli [1]. Most of the reported activatable prodrugs (>80%) are sensitive to a single trigger, including the tumor microenvironments (TMEs) like pH [2], glutathione (GSH) [3,4], reactive oxygen species (ROS) [5,6] and enzyme [7], or the external triggers like light, ultrasound, heat, and magnetic field [8,9]. However, in most of the cases, the nonspecific drug accumulation or activation in normal tissues is inevitable because some TMEs are not “specific” enough to sensitively differentiate tumor and normal tissues [10]. Applying “logic gate” involving multiple triggers into the molecular structures can remarkably magnify the specific tumor targeting and attenuate the undesired toxicity via the introduction of multistage “locks” and sophisticating the “unlock” conditions [11]. More than two stimuli co-govern the “on and off” of the drug, exhibiting “OR gate” or “AND gate” features [12]. Most of the reported prodrugs with logic gates are “OR gates”, that is, drugs can be partially activated by either stimulus [13,14]. Even though sometimes the tumor targeting and anti-cancer efficacy can be magnified upon the combination of multiple stimuli, the partial activation feature of the “OR gate” determines the toxicity induced by single stimulus cannot be thoroughly avoided [15]. Compared to “OR gate”, “AND gate” endows the prodrug with the “only” activation upon the co-existence of all the stimuli [16,17], which was first reported in the design of a boron dipyrromethene responsive to both Na+ and H+ ions by Akkaya [18]. However, due to the molecule synthesis and design challenging, the “AND gate” is infrequent among numerous prodrugs.
Among the stimuli, a combination of external stimulus and internal stimulus owns advanced strengths due to the tumor-specific and spatio-temporal control features. Ultrasound (US) as a mechanical force has been applied into the cancer treatment as an external stimulus, called sonodynamic therapy (SDT) [19]. Sonosensitizers bearing macrocyclic structures like porphyrin and phthalocyanine, can be activated under low-intensity US irradiation to produce highly toxic ROS through the sonoluminescence mechanism [20,21]. Compared to other treatments, SDT owns the advantages of spatio-temporal control, noninvasiveness, minor energy attenuation, and deep tissue-penetration [22,23]. However, the US-triggered “AND gate” prodrug has been rarely reported due to the following challenges. First, unlike the pro-chemodrug which the drug activation is based on the transformation or exposure of specific functional groups upon the triggers, it is hard to inhibit the sonosensitizers’ toxicity upon US trigger via simple change of functional groups [24,25]. As the toxicity of sonosensitizers is mainly originated from the US's energy and sonoluminescence, Förster resonance energy transfer (FRET) effect which has been widely applied in the design of dyes, is a promising strategy to suppress the toxicity from the “only” US irradiation [26]. Second, killing of the tumor cells for SDT greatly relies on the ROS (singlet oxygen (1O2) and oxygen-based radicals (•OH)) produced by the US-irradiated sonosensitizers. Limited by the tolerate dosage and intracellular uptake level of the sonosensitizers, as well as the oxidative ability and life time of the ROS, the anticancer efficacy for SDT cannot reach as good as that for chemotherapy or radiotherapy [27,28]. Therefore, stronger ROS agents or toxic agents are extremely needed to boost the cytotoxicity.
Considering the above-mentioned two issues in the design of “AND gate” SDT prodrugs, we specially design two monomers including a meso‑carboxyl-porphyrin-based sonosensitizer (5,10,15,20-tetrakis(carboxyl)porphyrin, TCP) and a croconium (thiophenyl-croconium (2,5-bis[(2-(2-(2-hydroxyethoxy)ethoxy)ethyl-4-carboxylate-piperidylamino)thiophenyl]-croconium, CR), as well as their polymers (pTCP-CR) to solve all the listed obstacles in a US and enzyme co-activated system. 5,10,15,20-Tetrakis(carboxyl)porphyrin (TCP) was selected since it can release ROS and carbon monoxide (CO) with US irradiation [29]. To further improve the toxicity of the ROS generated by SDT, sulfate radicals (SO4•−) with stronger oxidation capability are introduced into the molecule design. SO4•− has an oxidation potential of 2.5–3.1 eV, which is similar to that of •OH (2.80 eV), but the half-life (SO4•−, 30 µs) is 30 times versus •OH, which means that it has stronger cytotoxicity due to longer diffusion distance and leads to more effective oxidation reactions [30]. Recently, it has been revealed that SO4•− can be produced by sulfate ions (SO42−) and •OH, which is a mild method appliable in biomedical field [31]. Therefore, a specific CR dye bearing thiophene group is designed as a SO42− precursor since sulfur-containing dye owns the potential to release SO42− upon ROS treatment [32]. Third, the absorbance of the CR usually covers the emission of most porphyrin-based sonosensitizers upon US irradiation. Thus, CR can act as a quencher to lock the undesired US-induced ROS generated due to the FRET effect once it is connected with porphyrin via suitable linkers [26].
With the features of meso‑carboxyl porphyrin and thiophene-croconium at hand, TCP and CR are designed and polymerized into a polymeric prodrug (pTCP-CR) with cross-linked network structure via ester linkers (Scheme 1). Through self-assembly in water, it is prepared into nanoparticles (pTCP-CR NPs) (Scheme S2 in Supporting information) [33]. As shown in Scheme 1, activation of pTCP-CR NPs requires the presence of both carboxylesterase (CEs) and US. Under “only CEs” activation, pTCP-CR NPs cleave into TCP and CR monomers with low cytotoxicity. Under “only US” activation, ROS generation by TCP is quenched by CR, and CO generation is blocked by ester linkers. Only with the co-existence of CEs and US, TCP and CR release as monomers, enabling ROS and CO generation. ROS from TCP further decompose CR into SO42− and more toxic SO4•−, enhancing cytotoxicity. This enzyme and US co-triggered “AND gate” design enables precise spatio-temporal control over the release of anticancer agents. The efficacy of pTCP-CR NPs as a smart decomposable polysonosensitizer is demonstrated through in vitro experiments using cell lines with varying CEs expression levels and in vivo SDT efficacy on mice with hepatocellular carcinoma (HCC).
Although the instability and bleachability of squarylium dyes and CR dyes in the presence of metal ions or light have been reported [34], the phenomenon that CR dye can be degraded by ROS and transformed into active agents has not been reported. As sulfur-containing dyes owned the potentials to be converted to SO42− after being treated by ROS [32], CR bearing thiophenol group was selected as the special monomer for the releasing of surfur-based radicals. CR was synthesized by the nucleophilic substitution, alkali saponification/acid hydrolysis, condensation processes with croconic acid as shown in Scheme S2. All the intermediates and finally compounds were fully characterized (Figs. S1–S8 in Supporting information). It has been demonstrated before that US could trigger the porphyrin derivatives to generate •OH, similar with the porphyrin-based photodynamic therapy mechanism [35]. We then evaluated whether the ROS generated by the US and TCP would lead to the decomposition of CR into SO42− as well as more active SO4•− radicals (Fig. 1a). Adding •OH to a phosphate buffer saline (PBS) solution of CR led to a significant reduction in CR absorption in the near-infrared (NIR) region (around 700 nm) (Fig. 1b). 1H nuclear magnetic resonance spectroscopy (1H NMR) and high resolution mass spectrometry (HRMS) analyses confirmed CR decomposition after •OH treatment for 20 and 50 min, with CR molecules breaking down into low molecular weight compounds (Figs. S9 and S10 in Supporting information). Additionally, we assessed if •OH generated by TCP could decompose CR. As anticipated, CR absorption in the NIR region decreased after US treatment of a mixed solution of TCP and CR in PBS (Fig. S11 in Supporting information), while minimal change occurred in the CR + US group and the CR+TCP group (Fig. S11). This suggested CR decomposition relied on ROS from TCP and US combination. Furthermore, high performance high performance (HPIC) was utilized to determine SO42− concentration in various CR solutions (only CR group, CR + US group, CR + TCP + US group, and deionized water group). Results showed a significantly higher SO42− concentration in the CR + TCP + US group (3.88 mg/L) compared to control groups (0.5–0.7 mg/L) (Fig. 1d, Fig. S12 and Table S1 in Supporting information).
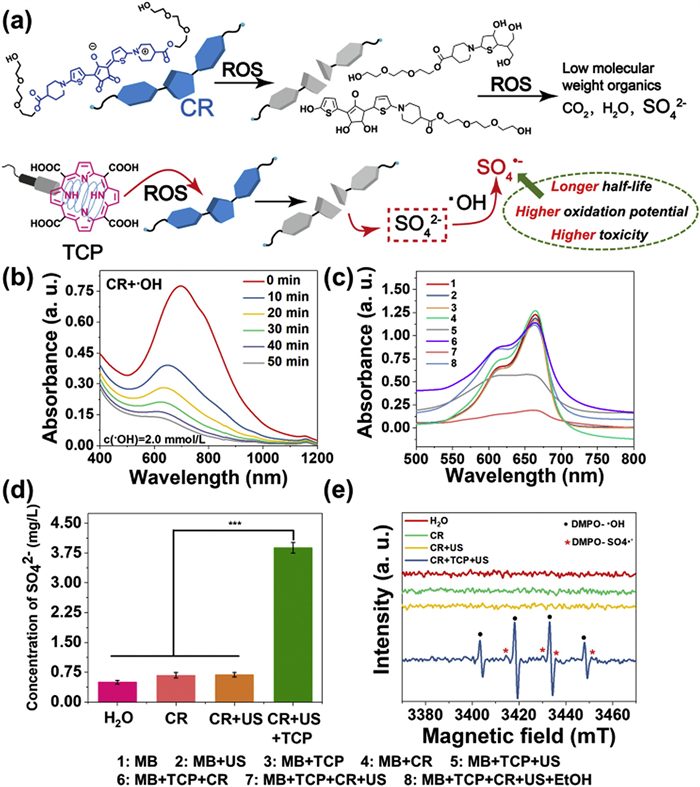
It has been reported that SO42− can be efficiently converted into SO4•− by •OH [36]. Electron paramagnetic resonance (EPR) analyses were used to examine the SO4•− radicals that are derived from both CR and TCP under US treatment (Fig. 1a). Obvious sulfate anion (DMPO-SO4•−, labeled by red star) signals were observed only in the CR + TCP + US treatment group (Fig. 1e). The EPR spectra supported that the •OH produced by TCP under US irradiation can effectively convert SO42− into SO4•−. Since the degradation of methylene blue (MB) could indirectly reflect the oxidative ability of •OH and SO4•−, the capability of •OH and SO4•− were compared using different combination of CR, TCP and US [37]. The results indicated that CR can effectively degrade MB in the presence of TCP and US, with up to 84.5% of MB being degraded, indicating that CR can produce •OH and SO4•− under the combined treatment of TCP and US (Fig. 1c and Fig. S13 in Supporting information).
Considering the advanced features of TCP and CR including the generation of CO, 1O2 and SO4•−, a polymerized TCP and CR (pTCP-CR) with cross-linked network structure connected by flexible ester linkers was designed and synthesized using our previously reported polycondensation method (Scheme S3 in Supporting information) [21,38]. As controls, polymerized TCPP and CR (pTCPP-CR) which could not release CO, and polymerized TCP with hexaethylene glycol (pTCP-HG) which did not generate SO4•− were also synthesized for comparisons. Their chemical structures were characterized by 1H NMR spectroscopy (Figs. S14–S16 in Supporting information). Their molecular weights (MWs) were calculated to be around 6900 (pTCP-CR), 9200 (pTCPP-CR) and 6100 (pTCP-HG) g/mol by gel permeation chromatography (GPC) (Fig. S17 in Supporting information). pTCP-CR, pTCPP-CR and pTCP-HG were prepared into nanoparticles (pTCP-CR NPs, pTCPP-CR NPs and pTCP NPs) in PBS by the self-assembly of hydrophilic and hydrophobic units [39,40]. These NPs showed unimodal size distributions with the average hydrodynamic diameters of 79.5 ± 2.8 nm (pTCP-CR NPs), 187.3 ± 3.3 nm (pTCPP-CR NPs) and 232.5 ± 6.2 nm (pTCP-HG NPs) measured by dynamic light scattering (DLS) (Fig. S18 in Supporting information). A spherical morphology of pTCP-CR NPs was observed with scanning electron microscopy (SEM) (Fig. S19 in Supporting information). After co-incubation with CEs (20 U/mL), the size of pTCP-CR NPs increased to over 1000 nm due to ester linker cleavage, causing nanoparticle structure destruction. These NPs contain three anti-cancer agents: CO, 1O2, and SO4•−, making them a prodrug. pTCP-CR NPs degrade into TCP and CR monomers when interacting with cancer cells overexpressing CEs but remain stable against normal tissues with limited CEs expression. TCP's emission (600–750 nm) overlaps with CR's absorption (600–800 nm), enabling the FRET effect (Figs. 2a, c and d) [41]. Therefore, the CR moiety in pTCP-CR NPs structure had the potential to act as a quencher, which would fully quench the fluorescence emission and ROS generation of TCP segment due to the presence of FRET process. However, the FRET effect of pTCP-CR NPs was inhibited in the monomeric state after CEs treatment (Fig. 2c and Figs. S22–S24 in Supporting information). The distance between the energy donor (TCP) and energy acceptor (CR) was an important factor affecting FRET effect [42]. The intermolecular interaction between TCP unit and CR unit is simulated. The intermolecular distance in the polymer mode (<10 Å) is much lower than that in the monomer mode (>15 Å) (Fig. S20 in Supporting information). In addition, due to the cleavage of pTCP-CR NPs by esterase, the change of FRET effect was time-dependent. In the polymer state, all the three active agents including CO, 1O2 and SO4•− were inactivated due to the quenching effect of CR on TCP.
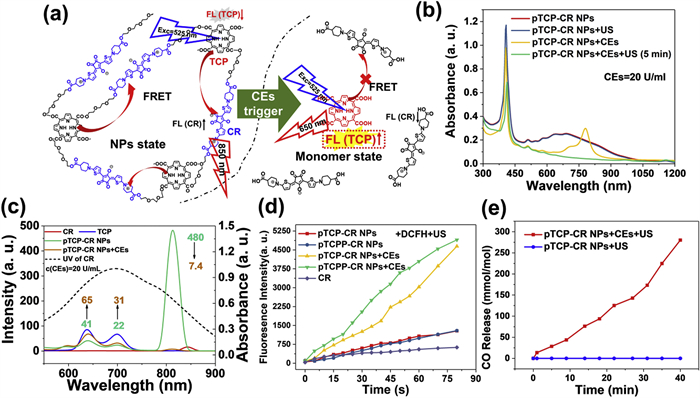
As a result, all the active agents were double-locked into pTCP-CR NPs, which could be only unlocked by the simultaneous existence of US and CEs (Scheme S4 in Supporting information). As shown in Fig. 2b, pTCP-CR NPs is processed by both US and CEs, both the TCP absorbance at 300–450 nm and CR absorbance at 600–1000 nm dramatically decreased. This indicates that both TCP and CR are decomposed after CEs/US dual processing. As a control, pTCP-CR NPs showed strong stability under only US or CEs irradiation (Fig. S25 in Supporting information). Subsequently, the ROS generation ability of CR, TCP + CR, pTCP-CR NPs and pTCPP-CR NPs in water upon different treatment conditions was evaluated. 1,3-Diphenylisobenzofuran (DPBF) and 2′,7′-dichlorodihyrofluorescein (DCFH) were used as 1O2 and ROS analytical reagent [43]. For CEs/US-co-treated pTCP-CR NPs and pTCPP-CR NPs, the absorption of DPBF (420 nm) decreased significantly with the prolongation of US treatment time, indicating the 1O2 generation upon US irradiation. As controls, if pTCP-CR NPs and pTCPP-CR NPs were only irradiation by US without CEs treatment, the absorption of DPBF did not change and no generation of 1O2 (Fig. S26 in Supporting information). Such results were in consistent with the fluorescence results that FRET effect inhibited the generation of 1O2 from TCP unit. Apart from DPBF, DCFH as a fluorescent probe, was also selected to demonstrate it. Compared with the only US triggered group, CEs/US co-triggered pTCP-CR NPs or pTCPP-CR NPs showed notable stronger fluorescence intensity, revealing the “on and off” of ROS was fully determined by the co-existence of CEs and US (Fig. 2d and Fig. S27 in Supporting information).
The CEs/US co-triggered CO release ability of pTCP-CR NPs was quantified by hemoglobin method, 9-(diethylamino)−5H-benzo[a]phenoxazin-5-one palladium complex (1-Ac) CO fluorescence probe method and CO gas sensor method. UV spectra indicated conversion of Hb-Fe(Ⅱ) to Hb-Fe(CO) after incubating CEs-treated pTCP-CR NPs with hemoglobin under US treatment. Fluorescence intensity of CEs-treated pTCP-CR NPs with 1-Ac increased significantly under US treatment, confirming CO release (Fig. S28 in Supporting information). CO gas sensor method quantified CO release at 280 mmol CO/mol TCP after 40 min of US treatment. No CO release was observed in pTCP-CR NPs treated only with US irradiation (Fig. 2e). Similar results were observed with pTCP-HG NPs (Fig. S28). Additionally, SO42− release from pTCP-CR NPs under CEs/US dual activation was significantly higher compared to other groups (Fig. S29 in Supporting information). In summary, the co-existence of CEs and US triggered the generation of CO, 1O2, and SO4•−.
Subsequently, we evaluated the in vitro release of ROS and CO from the double-blocked pTCP-CR NPs upon CEs/US triggers. It was known that the CEs expression in liver tumors was much higher than normal tissues [44]. CEs-positive hepatoma cells (HepG2) and CEs-negative cells (293T and LO2) were selected as the models to demonstrate the in vitro anticancer efficacy [45]. After incubation with pTCP-CR NPs (20 µmol/L porphyrin) for 4 h, bright fluorescence showed internalization in HepG2 cells, confirmed by colocalization with Mito-Tracker Green, indicating effective enrichment in mitochondria (Fig. 3a). Cellular uptake of pTCP-CR NPs was higher than free TCP in both HepG2 and 293T cells, suggesting improved internalization due to NPs formation [46]. Similar results were observed with pTCPP-CR NPs (Fig. S30 in Supporting information).
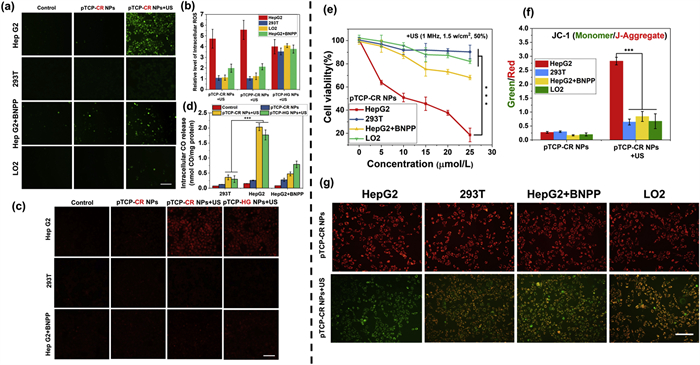
DCFH-diacetate (DA) and 1-Ac were furthermore used to detect the intracellular ROS and CO. In HepG2 cells, both pTCP-CR NPs and pTCPP-CR NPs showed green fluorescence upon US irradiation, while fluorescence dropped significantly in 293T and LO2 with or without US (Fig. 3a, Figs. S31 and S32 in Supporting information). Sole US treatment did not induce green fluorescence. The US-triggered ROS generation of pTCP-HG NPs was not influenced by cell type, blurring the tumor-normal cell distinction (Fig. S32). Adding CEs inhibitor bis-p-nitrophenyl phosphate (BNPP) to HepG2 cells reduced green fluorescence in pTCP-CR NPs + US and pTCPP-CR NPs + US groups, but not in pTCP-HG NPs + US group (Fig. 3b and Fig. S32). This aligns with ROS generation tuned by CEs and US co-existence, targeting toxicity to tumor cells under US. Similarly, intracellular CO release upon US showed CEs/US dependency. HepG2 cells incubated with pTCP-CR NPs and pTCP-HG NPs displayed intracellular CO production after US, evidenced by strong red fluorescence using 1-Ac (Figs. 3c and d, Fig. S33 in Supporting information). No CO was detected in 293T cells or HepG2 cells treated with BNPP. Thus, pTCP-CR NPs specifically generate ROS and CO only with CEs and US co-existence, targeting high CEs tumor cells under US.
The MTT assay evaluated the toxicity of pTCP-CR NPs, pTCPP-CR NPs, and pTCP-HG NPs on cancer cells. All NPs showed low cytotoxicity without US irradiation, indicating their safety (Fig. 3e and Fig. S34 Supporting information). Upon US irradiation, HepG2 cells treated with pTCP-CR NPs and pTCPP-CR NPs exhibited significantly higher cytotoxicity (Fig. 3f and Fig. S35 in Supporting information). Pre-treatment of HepG2 cells with BNPP decreased therapeutic efficiency, consistent with results in cells with limited CEs expression. At 25 µmol/L concentration (porphyrin equivalent), cell mortality was significantly higher in the pTCP-CR NPs + US group (81.37% ± 3.27%) compared to pTCPP-CR NPs + US (59.77% ± 1.35%) due to CO gas therapy from the TCP moiety (Fig. 3f). Additionally, mortality was higher in the pTCP-CR NPs + US group than pTCP-HG NPs + US (64.36% ± 2.25%) due to more efficient oxidative toxicity of SO4•− from CR (Fig. S36 in Supporting information). These results demonstrate the contribution of both SDT and GT therapy to cell death, activated only by CEs and US. CO and ROS can induce mitochondrial dysfunction and lead to cell apoptosis by decreasing mitochondrial membrane potential (MMP). To assess the therapeutic effect, MMP reduction was measured using 5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolo carbocyanine iodide (JC-1) dye. Following US-triggered treatment, HepG2 cells showed a significant decrease in MMP, shifting JC-1 dye fluorescence from red to green, indicating mitochondrial dysfunction (Fig. 3g). The green/red fluorescence intensity ratio, indicative of mitochondrial damage, was highest in HepG2 cells treated with pTCP-CR NPs and US, demonstrating the most effective mitochondrial damage compared to other cell lines (Figs. S36–S38 in Supporting information).
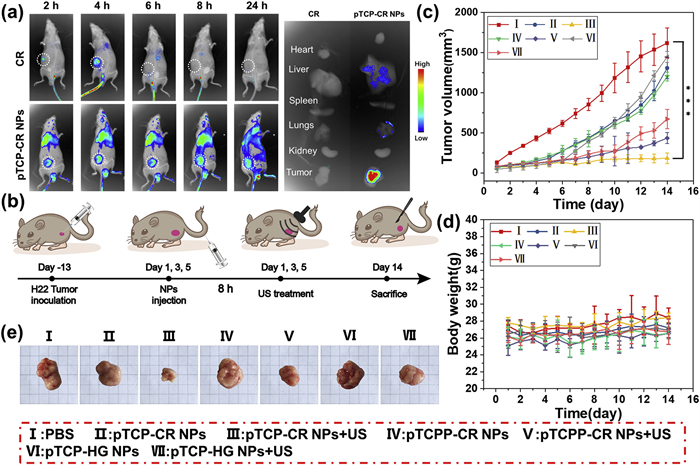
The in vivo synergistic anti-cancer therapy was furthermore evaluated using mice bearing H22 tumors. All animal experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of Dalian Medical University. The approval number of animal experiments is AEE22049. Initially, pTCP-CR NPs served as an in vivo fluorescence imaging agent to assess drug accumulation and retention. After intravenous injection into mice, fluorescence signals were recorded over time. Results showed discernible signals at the tumor site 2 h post-injection, peaking at 8 h. Minimal signals were observed in mice injected with free CR. Major organs and tumors were harvested 24 h post-injection for ex vivo fluorescence imaging (Fig. 4a). We evaluated the combined therapeutic efficacy of pTCP-CR NPs, pTCPP-CR NPs and pTCP-HG NPs in H22 tumor-bearing mice. Mice were divided into seven groups: PBS only, NPs only, NPs with US, with detailed administration in Fig. 4b, monitoring tumor sizes and body weights daily. Tumors in untreated and NPs-treated mice grew rapidly, while those in groups receiving US showed some inhibition compared to PBS. Notably, group Ⅲ (pTCP-CR NPs + US) displayed the highest inhibition, releasing both CO and enhanced ROS upon US. Tumor images and hematoxylin-eosin staining (H&E) staining confirmed these findings. Mice treated with NPs and US showed no significant weight loss or organ damage, suggesting safety (Figs. 4c–e, Figs. S39 and S40 in Supporting information).
In summary, a CEs and US co-triggered polysonosensitizer pTCP-CR NPs with “AND gate” logic was developed, in which TCP with ROS and CO generation ability was polymerized with CR with FRET quenching effect and SO4•− generation capability via ester linkers. Either CEs or US itself could not “unlock” pTCP-CR NPs with the releasing of active agents while the anti-cancer efficacy could only be activated upon the co-existence of CEs and US. Such design could avoid the nonspecific targeting and drug activation in the normal tissues via the combination of CEs as the internal trigger and US as an external trigger. A spatio-temporal control of the toxicity activation could also be achieved. Upon activation by two triggers, advanced anti-cancer agents beyond traditional ROS from SDT were simultaneously generated due to the smart design of TCP and CR monomers. TCP monomer can not only generate traditional ROS upon US irradiation, but also be decomposed into CO as therapeutic gas. CR monomer can not only quench the toxicity of TCP at polymer state due to FRET effect, but also be decomposed into SO4•− as more toxic ROS for toxicity boosting. Post SDT treatment, pTCP-CR NPs can be thoroughly decomposed into low-toxic molecules for the fast clearance, avoiding phototoxic side-ffect. Such “AND gate” logic design provided a promising strategy for the precise tumor treatment with limited side-effect.
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Shuxin Liu: Writing – review & editing, Writing – original draft, Data curation. Jinjuan Ma: Data curation. Aiguo Wang: Writing – review & editing, Writing – original draft, Resources. Nan Zheng: Writing – review & editing, Writing – original draft, Project administration, Conceptualization.
This work was supported by grants from the National Natural Science Foundation of China (No. 22375027), the Natural Science Foundation of Jiangsu Province (Nos. BK20221265, BK20211100), the Fundamental Research Funds for the Central Universities (No. DUT23YG133), and the Research Funds from Liaoning Cancer Hospital (No. 2024ZLKF-35). The authors acknowledge the assistance of DUT Instrumental Analysis Center.
Supplementary material associated with this article can be found, in the online version, at doi:
N.H. Tran, M. Reinhard, K.Y.H. Gin, Water Res. 133(2018) 182–207.
doi: 10.1016/j.watres.2017.12.029
P. Chowdhary, A. Raj, R.N. Bharagava, Chemosphere 194(2018) 229–246.
doi: 10.1016/j.chemosphere.2017.11.163
Z.N. Wang, M.Y. Liu, F. Xiao, et al., Chin. Chem. Lett. 33(2022) 653–662.
doi: 10.1016/j.cclet.2021.07.044
R. Khiewwijit, H. Temmink, H. Rijnaarts, K.J. Keesman, Environ. Model. Softw. 68(2015) 156–165.
doi: 10.1016/j.envsoft.2015.02.011
F.Q. Fan, R.H. Xu, D.P. Wang, F.G. Meng, Water Res. 181(2020) 115915.
doi: 10.1016/j.watres.2020.115915
Z.W. Zhang, Y.H. Wu, L.W. Luo, et al., Sci. Total Environ. 792(2021) 148291.
doi: 10.1016/j.scitotenv.2021.148291
M.N. Hasan, M.M. Altaf, N.A. Khan, et al., Chemosphere 277(2021) 130328.
doi: 10.1016/j.chemosphere.2021.130328
J.D. Xiao, Y.B. Xie, J. Rabeah, A. Brückner, H.B. Cao, Acc. Chem. Res. 53(2020) 1024–1033.
doi: 10.1021/acs.accounts.9b00624
R.C. Ji, J.B. Chen, T.C. Liu, X.F. Zhou, Y.L. Zhang, Chin. Chem. Lett. 33(2022) 643–652.
doi: 10.1016/j.cclet.2021.07.043
Y. Li, Y.G. Chen, J. Wu, Appl. Energy 240(2019) 120–137.
doi: 10.1016/j.apenergy.2019.01.243
H.Y. Wang, F. Qian, Y. Li, Nano Energy 8(2014) 264–273.
doi: 10.1016/j.nanoen.2014.06.004
T.H. Jeon, M.S. Koo, H. Kim, W. Choi, ACS Catal. 8(2018) 11542–11563.
doi: 10.1021/acscatal.8b03521
J. Highfield, Molecules 20(2015) 6739–6793.
doi: 10.3390/molecules20046739
H.J. Lewerenz, C. Heine, K. Skorupska, et al., Energy Environ. Sci. 3(2010) 748–760.
doi: 10.1039/b915922n
Z.D. Wei, J.Y. Liu, W.F. Shangguan, Chin. J. Catal. 41(2020) 1440–1450.
doi: 10.1016/S1872-2067(19)63448-0
Q. Wang, K. Domen, Chem. Rev. 120(2020) 919–985.
doi: 10.1021/acs.chemrev.9b00201
S. Kampouri, K.C. Stylianou, ACS Catal. 9(2019) 4247–4270.
doi: 10.1021/acscatal.9b00332
T.O. Ajiboye, O.A. Oyewo, D.C. Onwudiwe, Chemosphere 262(2021) 128379.
doi: 10.1016/j.chemosphere.2020.128379
L.M. Yang, W.B. Hu, Z.W. Chang, et al., Environ. Int. 152(2021) 106512.
doi: 10.1016/j.envint.2021.106512
L. Candish, K.D. Collins, G.C. Cook, et al., Chem. Rev. 122(2022) 2907–2980.
doi: 10.1021/acs.chemrev.1c00416
A. Fujishima, K. Honda, Nature 238(1972) 37–38.
doi: 10.1038/238037a0
Q. Guo, Z.B. Ma, C.Y. Zhou, Z.F. Ren, X.M. Yang, Chem. Rev. 119(2019) 11020–11041.
doi: 10.1021/acs.chemrev.9b00226
T.L. Xia, Y.C. Lin, W.Z. Li, M.T. Ju, Chin. Chem. Lett. 32(2021) 2975–2984.
doi: 10.1016/j.cclet.2021.02.058
F.E. Osterloh, ACS Energy Lett. 2(2017) 445–453.
doi: 10.1021/acsenergylett.6b00665
C.A. Martínez-Huitle, S. Ferro, Chem. Soc. Rev. 35(2006) 1324–1340.
doi: 10.1039/B517632H
G.Q. Zhao, Y.Z. Jiang, S.X. Dou, W.P. Sun, H.G. Pan, Sci. Bull. 66(2021) 85–96.
doi: 10.1016/j.scib.2020.09.014
H.X. Zhong, M.C. Wang, G.B. Chen, R.H. Dong, X.L. Feng, ACS Nano 16(2022) 1759–1780.
doi: 10.1021/acsnano.1c10544
J. Linnemann, K. Kanokkanchana, K. Tschulik, ACS Catal. 11(2021) 5318–5346.
doi: 10.1021/acscatal.0c04118
L. Rebollar, S. Intikhab, N.J. Oliveira, et al., ACS Catal. 10(2020) 14747–14762.
doi: 10.1021/acscatal.0c03801
X.V. Medvedeva, J.J. Medvedev, S.W. Tatarchuk, R.M. Choueiri, A. Klinkova, Green Chem. 22(2020) 4456–4462.
doi: 10.1039/D0GC01754J
Y.L. Quan, J.X. Zhu, G.F. Zheng, Small 1(2021) 2100043.
doi: 10.1002/smsc.202100043
D. Shahidi, R. Roy, A. Azzouz, Appl. Catal. B: Environ. 174(2015) 277–292.
S.Z. Xu, E.A. Carter, Chem. Rev. 119(2019) 6631–6669.
doi: 10.1021/acs.chemrev.8b00481
B.M. Tackett, E. Gomez, J.G. Chen, Nat. Catal. 2(2019) 381–386.
doi: 10.1038/s41929-019-0266-y
P. Prabhu, V. Jose, J.M. Lee, Adv. Funct. Mater. 30(2020) 1910768.
doi: 10.1002/adfm.201910768
X.X. Chang, T. Wang, J.L. Gong, Energy Environ. Sci. 9(2016) 2177–2196.
doi: 10.1039/C6EE00383D
D.N. Jiang, P. Xu, H. Wang, et al., Coordin. Chem. Rev. 376(2018) 449–466.
doi: 10.1016/j.ccr.2018.08.005
Z.J. Wang, H. Song, H.M. Liu, J.H. Ye, Angew. Chem. Int. Ed. 59(2020) 8016–8035.
doi: 10.1002/anie.201907443
M. Melchionna, P. Fornasiero, ACS Catal. 10(2020) 5493–5501.
doi: 10.1021/acscatal.0c01204
J.P. Zou, D.D. Wu, J.M. Luo, et al., ACS Catal. 6(2016) 6861–6867.
doi: 10.1021/acscatal.6b01729
G. Rossi, L. Pasquini, D. Catone, et al., Appl. Catal. B: Environ. 237(2018) 603–612.
doi: 10.1016/j.apcatb.2018.06.011
Y.B. Yan, J. Gong, J. Chen, et al., Adv. Mater. 31(2019) 1808283.
doi: 10.1002/adma.201808283
V.C. Hoang, K. Dave, V.G. Gomes, Nano Energy 66(2019) 104093.
doi: 10.1016/j.nanoen.2019.104093
Y. Yamaguchi, A. Kudo, Front. Energy 15(2021) 568–576.
doi: 10.1007/s11708-021-0774-8
R. Acharya, K. Parida, J. Environ. Chem. Eng. 8(2020) 103896.
doi: 10.1016/j.jece.2020.103896
W.H. Dong, D.D. Wu, J.M. Luo, et al., J. Catal. 349(2017) 218–225.
doi: 10.1016/j.jcat.2017.02.004
X. Lu, C.Q. Zhu, Z.S. Wu, et al., J. Am. Chem. Soc. 142(2020) 15438–15444.
doi: 10.1021/jacs.0c06779
D.A. Henckel, M.J. Counihan, H.E. Holmes, et al., ACS Catal. 11(2021) 255–263.
doi: 10.1021/acscatal.0c04297
Q. Xie, W.M. He, S.W. Liu, Chin. J. Catal. 41(2020) 140–153.
doi: 10.1016/S1872-2067(19)63481-9
M.F.R. Samsudin, H. Ullah, R. Bashiri, et al., ACS Sustain. Chem. Eng. 8(2020) 9393–9403.
doi: 10.1021/acssuschemeng.0c02063
Y. Wang, G.Q. Tan, T. Liu, et al., Appl. Catal. B: Environ. 234(2018) 37–49.
doi: 10.1016/j.apcatb.2018.04.026
R.Z. Zhang, B.Y. Wu, Q. Li, et al., Coordin. Chem. Rev. 422(2020) 213436.
doi: 10.1016/j.ccr.2020.213436
L. Zhang, Z.J. Zhao, T. Wang, J.L. Gong, Chem. Soc. Rev. 47(2018) 5423–5443.
doi: 10.1039/C8CS00016F
Z.W. Seh, J. Kibsgaard, C.F. Dickens, et al., Science 355(2017) eaad4998.
doi: 10.1126/science.aad4998
F. Franco, C. Rettenmaier, H.S. Jeon, B.R. Cuenya, Chem. Soc. Rev. 49(2020) 6884–6946.
doi: 10.1039/D0CS00835D
Q. Lu, J. Rosen, Y. Zhou, et al., Nat. Commun. 5(2014) 3242.
doi: 10.1038/ncomms4242
J. Rosen, G.S. Hutchings, Q. Lu, et al., ACS Catal. 5(2015) 4586–4591.
doi: 10.1021/acscatal.5b00922
D.S. Ripatti, T.R. Veltman, M.W. Kanan, Joule 3(2019) 240–256.
doi: 10.1016/j.joule.2018.10.007
S. Verma, X. Lu, S.C. Ma, R.I. Masel, P.J. Kenis, Phys. Chem. Chem. Phys. 18(2016) 7075–7084.
doi: 10.1039/C5CP05665A
D.M. Weekes, D.A. Salvatore, A. Reyes, A. Huang, C. Berlinguette, Acc. Chem. Res. 51(2018) 910–918.
doi: 10.1021/acs.accounts.8b00010
G. Bharath, K. Rambabu, C. Aubry, et al., ACS Appl. Energy Mater. 4(2021) 11408–11418.
doi: 10.1021/acsaem.1c02196
Y.K. Long, J. Dai, S.Y. Zhao, et al., Environ. Sci. Technol. 55(2021) 5357–5370.
doi: 10.1021/acs.est.0c07794
Y.L. Chen, X. Bai, Y.T. Ji, T. Shen, Chem. Eng. J. 430(2022) 132951.
doi: 10.1016/j.cej.2021.132951
L. Wang, J.W. Wan, Y.S. Zhao, N.L. Yang, D. Wang, J. Am. Chem. Soc. 141(2019) 2238–2241.
doi: 10.1021/jacs.8b13528
W.C. Lai, Z.S. Ma, J.W. Zhang, et al., Adv. Func. Mater. 32(2022) 2111193.
doi: 10.1002/adfm.202111193
Q.N. Wang, X.Q. Wang, C. Wu, Y.Y. Cheng, Q.Y. Sun, H.B. Yu, J. CO2 Util. 26(2018) 425–433.
doi: 10.1016/j.jcou.2018.05.027
T.N. Nguyen, C.T. Dinh, Chem. Soc. Rev. 49(2020) 7488–7504.
doi: 10.1039/D0CS00230E
H. Rabiee, L. Ge, X.Q. Zhang, et al., Energy Environ. Sci. 14(2021) 1959–2008.
doi: 10.1039/D0EE03756G
H. Dong, M. Lu, Y. Wang, et al., Appl. Catal. B: Environ. 303(2022) 120897.
doi: 10.1016/j.apcatb.2021.120897
M.L. Zhang, Z.D. Zhang, Z.H. Zhao, et al., ACS Catal. 11(2021) 11103–11108.
doi: 10.1021/acscatal.1c02556
W.F. Xie, H. Li, G.Q. Cui, et al., Angew. Chem. Int. Ed. 60(2021) 7382–7388.
doi: 10.1002/anie.202014655
Q.N. Wang, C.Q. Zhu, C. Wu, H.B. Yu, Electrochim. Acta 319(2019) 138–147.
doi: 10.1016/j.electacta.2019.06.167
J. Wu, K. Zhu, H. Xu, W. Yan, Chin. J. Catal. 40(2019) 917–927.
doi: 10.1016/S1872-2067(19)63342-5
N. Nandal, S.L. Jain, Coordin. Chem. Rev. 451(2022) 214271.
doi: 10.1016/j.ccr.2021.214271
E. Kusmierek, Catalysts 10(2020) 439.
doi: 10.3390/catal10040439
S.S. Liu, Q.J. Xing, Y. Chen, et al., ACS Sustain. Chem. Eng. 7(2019) 1250–1259.
doi: 10.1021/acssuschemeng.8b04917
J. Zhang, S. Lv, J.L. Zheng, et al., ACS Sustain. Chem. Eng. 8(2020) 11133–11140.
doi: 10.1021/acssuschemeng.0c01906
J.F. de Brito, J.A.L. Perini, S. Perathoner, M.V.B. Zanoni, Electrochim. Acta 306(2019) 277–284.
doi: 10.1016/j.electacta.2019.03.134
D. Wang, Y.N. He, N. Zhong, et al., J. Hazard. Mater. 410(2021) 124563.
doi: 10.1016/j.jhazmat.2020.124563
D.B. Miklos, C. Remy, M. Jekel, et al., Water Res. 139(2018) 118–131.
doi: 10.1016/j.watres.2018.03.042
X.G. Duan, H.Q. Sun, S.B. Wang, Acc. Chem. Res. 51(2018) 678–687.
doi: 10.1021/acs.accounts.7b00535
J. Lee, U. von Gunten, J.H. Kim, Environ. Sci. Technol. 54(2020) 3064–3081.
doi: 10.1021/acs.est.9b07082
J.P. Zou, Y. Chen, S.S. Liu, et al., Water Res. 150(2019) 330–339.
doi: 10.1016/j.watres.2018.11.077
W.D. Oh, Z.L. Dong, T.T. Lim, Appl. Catal. B: Environ. 194(2016) 169–201.
doi: 10.1016/j.apcatb.2016.04.003
R. Cheula, M. Maestri, G. Mpourmpakis, ACS Catal. 10(2020) 6149–6158.
doi: 10.1021/acscatal.0c01005
H.Y. Tan, J. Wang, S.Z. Yu, K.B. Zhou, Environ. Sci. Technol. 49(2015) 8675–8682.
doi: 10.1021/acs.est.5b01264
M. Zhu, L.S. Zhang, S.S. Liu, et al., Chin. Chem. Lett. 31(2020) 1961–1965.
doi: 10.1016/j.cclet.2020.01.017
J. Wang, W. Liu, D.M. Li, Y.P. Wang, J. Alloy. Compd. 588(2014) 378–383.
doi: 10.1016/j.jallcom.2013.11.040
Z. Wang, C. Li, K. Domen, Chem. Soc. Rev. 48(2019) 2109–2125.
doi: 10.1039/C8CS00542G
L. Wang, X.L. Geng, L. Zhang, et al., Chemosphere 286(2022) 131558.
doi: 10.1016/j.chemosphere.2021.131558
C.G. Liu, Z.F. Lei, Y.N. Yang, Z.Y. Zhang, Water Res. 47(2013) 49586–49592.
X.J. Zheng, C.L. Li, M. Zhao, et al., Int. J. Hydrog. Energy 42(2017) 7917–7929.
doi: 10.1016/j.ijhydene.2016.12.131
Y. Rong, L. Tang, Y.H. Song, et al., RSC Adv. 6(2016) 80595–80603.
doi: 10.1039/C6RA15320H
K.H. Chu, L.Q. Ye, W. Wang, et al., Chemosphere 183(2016) 219–228.
L. He, L. Li, T.T. Wang, et al., Dalton Trans. 43(2014) 16981–16985.
doi: 10.1039/C4DT02557A
R. Shwetharani, M. Sakar, H.R. Chandan, R.G. Balakrishna, Mater. Lett. 218(2018) 262–265.
doi: 10.1016/j.matlet.2018.02.031
Y.P. Peng, H.L. Chen, C.P. Huang, Appl. Catal. B: Environ. 209(2017) 437–446.
doi: 10.1016/j.apcatb.2017.02.084
J. Han, Y.R. Bian, X.Z. Zheng, X.M. Sun, L.W. Zhang, Chin. Chem. Lett. 28(2017) 2239–2243.
doi: 10.1016/j.cclet.2017.08.031
A. Patsoura, D.I. Kondarides, X.E. Verykios, Appl. Catal. B: Environ. 64(2006) 171–179.
doi: 10.1016/j.apcatb.2005.11.015
J.G. Wang, P. Zhang, X. Li, J. Zhu, H.X. Li, Appl. Catal. B: Environ. 134-135(2013) 198–204.
doi: 10.1016/j.apcatb.2013.01.006
M.Q. Hu, Z.P. Xing, Y. Cao, et al., Appl. Catal. B: Environ. 226(2018) 499–508.
doi: 10.1016/j.apcatb.2017.12.069
K.X. Li, Z.X. Zeng, L.S. Yan, et al., Appl. Catal. B: Environ. 187(2016) 269–280.
doi: 10.1016/j.apcatb.2016.01.046
H. Park, A. Bak, Y.Y. Ahn, J. Choi, M.R. Hoffmannn, J. Hazard. Mater. 211-212(2012) 47–54.
doi: 10.1016/j.jhazmat.2011.05.009
Y.C. Nie, F. Yu, L.C. Wang, et al., Appl. Catal. B: Environ. 227(2018) 312–321.
doi: 10.1016/j.apcatb.2018.01.033
X.H. Jiang, L.C. Wang, F. Yu, et al., ACS Sustain. Chem. Eng. 6(2018) 12695–12705.
doi: 10.1021/acssuschemeng.8b01695
J.P. Zou, D.D. Wu, S.K. Bao, et al., ACS Appl. Mater. Interfaces 7(2015) 28429–28437.
doi: 10.1021/acsami.5b09255
D.K. Wang, H. Zeng, X. Xiong, et al., Sci. Bull. 65(2020) 113–122.
doi: 10.1016/j.scib.2019.10.015
F. Lu, D. Astruc, Coordin. Chem. Rev. 356(2018) 147–164.
doi: 10.1016/j.ccr.2017.11.003
K. Vikrant, K.H. Kim, Chem. Eng. J. 358(2019) 264–282.
doi: 10.1016/j.cej.2018.10.022
J.J. Rueda-Marquez, I. Levchuk, P.F. Ibanez, M. Sillanpaa, J. Clean. Prod. 258(2020) 120694.
doi: 10.1016/j.jclepro.2020.120694
S. Al-Amshawee, M.Y.B.M. Yunus, A.A.M. Azoddein, et al., Chem. Eng. J. 380(2020) 122231.
doi: 10.1016/j.cej.2019.122231
G.R. Xu, Z.H. An, K. Xu, et al., Coordin. Chem. Rev. 427(2021) 213554.
doi: 10.1016/j.ccr.2020.213554
Y. Zhu, W.H. Fan, T.T. Zhou, X.M. Li, Sci. Total Environ. 678(2019) 253–266.
doi: 10.1016/j.scitotenv.2019.04.416
Z. Xu, Q.R. Zhang, X.C. Li, X.F. Huang, Chem. Eng. J. 429(2022) 131688.
doi: 10.1016/j.cej.2021.131688
X. Zhao, L.B. Guo, B.F. Zhang, H.J. Liu, J.H. Qu, Environ. Sci. Technol. 47(2013) 4480–4488.
doi: 10.1021/es3046982
F. Zhang, W.L. Wang, C.Z. Zhou, Y.L. Sun, J.F. Niu, Chemosphere 278(2021) 130465.
doi: 10.1016/j.chemosphere.2021.130465
H.B. Zeng, S.S. Liu, B.Y. Chai, et al., Environ. Sci. Technol. 50(2016) 6459–6466.
doi: 10.1021/acs.est.6b00632
Z H, W.Q. Guo Wang, B.H. Liu, et al., Water Res. 160(2019) 405–414.
doi: 10.1016/j.watres.2019.05.059
D.K. Wang, H. Zeng, S.Q. Chen, J. Catal. 406(2022) 1–8.
doi: 10.1016/j.jcat.2021.12.027
L. Tian, P. Chen, X.H. Jiang, et al., Water Res. 209(2022) 117890.
doi: 10.1016/j.watres.2021.117890
S.C. Tian, C.Z. Dang, R. Mao, X. Zhao, ACS Sustain. Chem. Eng. 6(2018) 10273–10281.
doi: 10.1021/acssuschemeng.8b01634
K. Xiao, B. Zhou, S.Y. Chen, et al., Electrochem. Commun. 100(2019) 34–38.
doi: 10.1016/j.elecom.2019.01.018
X. Zhao, J.J. Zhang, M. Qiao, H.J. Liu, J.H. Qu, Environ. Sci. Technol. 49(2015) 4567–4574.
doi: 10.1021/es5062374
K.H. Xue, J. Wang, R. He, et al., Sci. Total Environ. 732(2020) 138963.
doi: 10.1016/j.scitotenv.2020.138963
X.H. Jiang, Q.J. Xing, X.B. Luo, et al., Appl. Catal. B: Environ. 228(2018) 29–38.
doi: 10.1016/j.apcatb.2018.01.062
J.M. Luo, S.Q. Zhang, M. Sun, et al., ACS Nano 13(2019) 9811–9840.
doi: 10.1021/acsnano.9b03649
Y.Y. Wu, Y.Q. Li, H.J. Hu, G.S. Zeng, C.H. Li, ACS ES & T Eng. 1(2021) 603–611.
X.X. Ma, X.K. Liu, J.H. Tang, et al., Appl. Surf. Sci. 602(2022) 154276.
doi: 10.1016/j.apsusc.2022.154276
M.S. Koo, X.F. Chen, K. Cho, T.C. An, W. Choi, Environ. Sci. Technol. 53(2019) 9926–9936.
doi: 10.1021/acs.est.9b02401
X.H. Jiang, L.S. Zhang, H.Y. Liu, et al., Angew. Chem. Int. Ed. 59(2020) 23112–23116.
doi: 10.1002/anie.202011495
L.S. Zhang, X.H. Jiang, Z.A. Zhong, et al., Angew. Chem. Int. Ed. 60(2021) 21751–21755.
doi: 10.1002/anie.202109488
M. Govindan, K.C. Pillai, B. Subramanian, I.S. Moon, ACS Omega 2(2017) 3562–3571.
doi: 10.1021/acsomega.7b00352
J. Kim, W. Choi, Energy Environ. Sci. 6(2010) 1042–1045.
J. Kim, D. Monllor-Satoca, W. Choi, Energy Environ. Sci. 5(2012) 7647–7656.
doi: 10.1039/c2ee21310a
G. Iervolino, V. Vaiano, J.J. Murcia, et al., J. Catal. 339(2016) 47–56.
doi: 10.1016/j.jcat.2016.03.032
D. Monllor-Satoca, R. Gómez, J. Phys. Chem. C 112(2008) 139–147.
doi: 10.1021/jp075672r
A. Heuer-Jungemann, N. Feliu, I. Bakaimi, et al., Chem. Rev. 119(2019) 4819–4880.
doi: 10.1021/acs.chemrev.8b00733
G. Hippargi, S. Anjankar, R.J. Krupadam, S.S. Rayalu, Fuel 291(2021) 120113.
doi: 10.1016/j.fuel.2020.120113
N. Sun, Y. Qu, C.H. Yang, et al., Appl. Catal. B: Environ. 263(2020) 118313.
doi: 10.1016/j.apcatb.2019.118313
Z.Y. Wu, Z.Y. Zhou, Y.J. Zhang, et al., Electrochim. Acta 254(2017) 140–147.
doi: 10.1016/j.electacta.2017.09.130
S.Q. Zhang, L.L. Wang, C.B. Liu, et al., Water Res. 121(2017) 11–19.
doi: 10.1016/j.watres.2017.05.013
N.H. Tran, M. Reinhard, K.Y.H. Gin, Water Res. 133(2018) 182–207.
doi: 10.1016/j.watres.2017.12.029
P. Chowdhary, A. Raj, R.N. Bharagava, Chemosphere 194(2018) 229–246.
doi: 10.1016/j.chemosphere.2017.11.163
Z.N. Wang, M.Y. Liu, F. Xiao, et al., Chin. Chem. Lett. 33(2022) 653–662.
doi: 10.1016/j.cclet.2021.07.044
R. Khiewwijit, H. Temmink, H. Rijnaarts, K.J. Keesman, Environ. Model. Softw. 68(2015) 156–165.
doi: 10.1016/j.envsoft.2015.02.011
F.Q. Fan, R.H. Xu, D.P. Wang, F.G. Meng, Water Res. 181(2020) 115915.
doi: 10.1016/j.watres.2020.115915
Z.W. Zhang, Y.H. Wu, L.W. Luo, et al., Sci. Total Environ. 792(2021) 148291.
doi: 10.1016/j.scitotenv.2021.148291
M.N. Hasan, M.M. Altaf, N.A. Khan, et al., Chemosphere 277(2021) 130328.
doi: 10.1016/j.chemosphere.2021.130328
J.D. Xiao, Y.B. Xie, J. Rabeah, A. Brückner, H.B. Cao, Acc. Chem. Res. 53(2020) 1024–1033.
doi: 10.1021/acs.accounts.9b00624
R.C. Ji, J.B. Chen, T.C. Liu, X.F. Zhou, Y.L. Zhang, Chin. Chem. Lett. 33(2022) 643–652.
doi: 10.1016/j.cclet.2021.07.043
Y. Li, Y.G. Chen, J. Wu, Appl. Energy 240(2019) 120–137.
doi: 10.1016/j.apenergy.2019.01.243
H.Y. Wang, F. Qian, Y. Li, Nano Energy 8(2014) 264–273.
doi: 10.1016/j.nanoen.2014.06.004
T.H. Jeon, M.S. Koo, H. Kim, W. Choi, ACS Catal. 8(2018) 11542–11563.
doi: 10.1021/acscatal.8b03521
J. Highfield, Molecules 20(2015) 6739–6793.
doi: 10.3390/molecules20046739
H.J. Lewerenz, C. Heine, K. Skorupska, et al., Energy Environ. Sci. 3(2010) 748–760.
doi: 10.1039/b915922n
Z.D. Wei, J.Y. Liu, W.F. Shangguan, Chin. J. Catal. 41(2020) 1440–1450.
doi: 10.1016/S1872-2067(19)63448-0
Q. Wang, K. Domen, Chem. Rev. 120(2020) 919–985.
doi: 10.1021/acs.chemrev.9b00201
S. Kampouri, K.C. Stylianou, ACS Catal. 9(2019) 4247–4270.
doi: 10.1021/acscatal.9b00332
T.O. Ajiboye, O.A. Oyewo, D.C. Onwudiwe, Chemosphere 262(2021) 128379.
doi: 10.1016/j.chemosphere.2020.128379
L.M. Yang, W.B. Hu, Z.W. Chang, et al., Environ. Int. 152(2021) 106512.
doi: 10.1016/j.envint.2021.106512
L. Candish, K.D. Collins, G.C. Cook, et al., Chem. Rev. 122(2022) 2907–2980.
doi: 10.1021/acs.chemrev.1c00416
A. Fujishima, K. Honda, Nature 238(1972) 37–38.
doi: 10.1038/238037a0
Q. Guo, Z.B. Ma, C.Y. Zhou, Z.F. Ren, X.M. Yang, Chem. Rev. 119(2019) 11020–11041.
doi: 10.1021/acs.chemrev.9b00226
T.L. Xia, Y.C. Lin, W.Z. Li, M.T. Ju, Chin. Chem. Lett. 32(2021) 2975–2984.
doi: 10.1016/j.cclet.2021.02.058
F.E. Osterloh, ACS Energy Lett. 2(2017) 445–453.
doi: 10.1021/acsenergylett.6b00665
C.A. Martínez-Huitle, S. Ferro, Chem. Soc. Rev. 35(2006) 1324–1340.
doi: 10.1039/B517632H
G.Q. Zhao, Y.Z. Jiang, S.X. Dou, W.P. Sun, H.G. Pan, Sci. Bull. 66(2021) 85–96.
doi: 10.1016/j.scib.2020.09.014
H.X. Zhong, M.C. Wang, G.B. Chen, R.H. Dong, X.L. Feng, ACS Nano 16(2022) 1759–1780.
doi: 10.1021/acsnano.1c10544
J. Linnemann, K. Kanokkanchana, K. Tschulik, ACS Catal. 11(2021) 5318–5346.
doi: 10.1021/acscatal.0c04118
L. Rebollar, S. Intikhab, N.J. Oliveira, et al., ACS Catal. 10(2020) 14747–14762.
doi: 10.1021/acscatal.0c03801
X.V. Medvedeva, J.J. Medvedev, S.W. Tatarchuk, R.M. Choueiri, A. Klinkova, Green Chem. 22(2020) 4456–4462.
doi: 10.1039/D0GC01754J
Y.L. Quan, J.X. Zhu, G.F. Zheng, Small 1(2021) 2100043.
doi: 10.1002/smsc.202100043
D. Shahidi, R. Roy, A. Azzouz, Appl. Catal. B: Environ. 174(2015) 277–292.
S.Z. Xu, E.A. Carter, Chem. Rev. 119(2019) 6631–6669.
doi: 10.1021/acs.chemrev.8b00481
B.M. Tackett, E. Gomez, J.G. Chen, Nat. Catal. 2(2019) 381–386.
doi: 10.1038/s41929-019-0266-y
P. Prabhu, V. Jose, J.M. Lee, Adv. Funct. Mater. 30(2020) 1910768.
doi: 10.1002/adfm.201910768
X.X. Chang, T. Wang, J.L. Gong, Energy Environ. Sci. 9(2016) 2177–2196.
doi: 10.1039/C6EE00383D
D.N. Jiang, P. Xu, H. Wang, et al., Coordin. Chem. Rev. 376(2018) 449–466.
doi: 10.1016/j.ccr.2018.08.005
Z.J. Wang, H. Song, H.M. Liu, J.H. Ye, Angew. Chem. Int. Ed. 59(2020) 8016–8035.
doi: 10.1002/anie.201907443
M. Melchionna, P. Fornasiero, ACS Catal. 10(2020) 5493–5501.
doi: 10.1021/acscatal.0c01204
J.P. Zou, D.D. Wu, J.M. Luo, et al., ACS Catal. 6(2016) 6861–6867.
doi: 10.1021/acscatal.6b01729
G. Rossi, L. Pasquini, D. Catone, et al., Appl. Catal. B: Environ. 237(2018) 603–612.
doi: 10.1016/j.apcatb.2018.06.011
Y.B. Yan, J. Gong, J. Chen, et al., Adv. Mater. 31(2019) 1808283.
doi: 10.1002/adma.201808283
V.C. Hoang, K. Dave, V.G. Gomes, Nano Energy 66(2019) 104093.
doi: 10.1016/j.nanoen.2019.104093
Y. Yamaguchi, A. Kudo, Front. Energy 15(2021) 568–576.
doi: 10.1007/s11708-021-0774-8
R. Acharya, K. Parida, J. Environ. Chem. Eng. 8(2020) 103896.
doi: 10.1016/j.jece.2020.103896
W.H. Dong, D.D. Wu, J.M. Luo, et al., J. Catal. 349(2017) 218–225.
doi: 10.1016/j.jcat.2017.02.004
X. Lu, C.Q. Zhu, Z.S. Wu, et al., J. Am. Chem. Soc. 142(2020) 15438–15444.
doi: 10.1021/jacs.0c06779
D.A. Henckel, M.J. Counihan, H.E. Holmes, et al., ACS Catal. 11(2021) 255–263.
doi: 10.1021/acscatal.0c04297
Q. Xie, W.M. He, S.W. Liu, Chin. J. Catal. 41(2020) 140–153.
doi: 10.1016/S1872-2067(19)63481-9
M.F.R. Samsudin, H. Ullah, R. Bashiri, et al., ACS Sustain. Chem. Eng. 8(2020) 9393–9403.
doi: 10.1021/acssuschemeng.0c02063
Y. Wang, G.Q. Tan, T. Liu, et al., Appl. Catal. B: Environ. 234(2018) 37–49.
doi: 10.1016/j.apcatb.2018.04.026
R.Z. Zhang, B.Y. Wu, Q. Li, et al., Coordin. Chem. Rev. 422(2020) 213436.
doi: 10.1016/j.ccr.2020.213436
L. Zhang, Z.J. Zhao, T. Wang, J.L. Gong, Chem. Soc. Rev. 47(2018) 5423–5443.
doi: 10.1039/C8CS00016F
Z.W. Seh, J. Kibsgaard, C.F. Dickens, et al., Science 355(2017) eaad4998.
doi: 10.1126/science.aad4998
F. Franco, C. Rettenmaier, H.S. Jeon, B.R. Cuenya, Chem. Soc. Rev. 49(2020) 6884–6946.
doi: 10.1039/D0CS00835D
Q. Lu, J. Rosen, Y. Zhou, et al., Nat. Commun. 5(2014) 3242.
doi: 10.1038/ncomms4242
J. Rosen, G.S. Hutchings, Q. Lu, et al., ACS Catal. 5(2015) 4586–4591.
doi: 10.1021/acscatal.5b00922
D.S. Ripatti, T.R. Veltman, M.W. Kanan, Joule 3(2019) 240–256.
doi: 10.1016/j.joule.2018.10.007
S. Verma, X. Lu, S.C. Ma, R.I. Masel, P.J. Kenis, Phys. Chem. Chem. Phys. 18(2016) 7075–7084.
doi: 10.1039/C5CP05665A
D.M. Weekes, D.A. Salvatore, A. Reyes, A. Huang, C. Berlinguette, Acc. Chem. Res. 51(2018) 910–918.
doi: 10.1021/acs.accounts.8b00010
G. Bharath, K. Rambabu, C. Aubry, et al., ACS Appl. Energy Mater. 4(2021) 11408–11418.
doi: 10.1021/acsaem.1c02196
Y.K. Long, J. Dai, S.Y. Zhao, et al., Environ. Sci. Technol. 55(2021) 5357–5370.
doi: 10.1021/acs.est.0c07794
Y.L. Chen, X. Bai, Y.T. Ji, T. Shen, Chem. Eng. J. 430(2022) 132951.
doi: 10.1016/j.cej.2021.132951
L. Wang, J.W. Wan, Y.S. Zhao, N.L. Yang, D. Wang, J. Am. Chem. Soc. 141(2019) 2238–2241.
doi: 10.1021/jacs.8b13528
W.C. Lai, Z.S. Ma, J.W. Zhang, et al., Adv. Func. Mater. 32(2022) 2111193.
doi: 10.1002/adfm.202111193
Q.N. Wang, X.Q. Wang, C. Wu, Y.Y. Cheng, Q.Y. Sun, H.B. Yu, J. CO2 Util. 26(2018) 425–433.
doi: 10.1016/j.jcou.2018.05.027
T.N. Nguyen, C.T. Dinh, Chem. Soc. Rev. 49(2020) 7488–7504.
doi: 10.1039/D0CS00230E
H. Rabiee, L. Ge, X.Q. Zhang, et al., Energy Environ. Sci. 14(2021) 1959–2008.
doi: 10.1039/D0EE03756G
H. Dong, M. Lu, Y. Wang, et al., Appl. Catal. B: Environ. 303(2022) 120897.
doi: 10.1016/j.apcatb.2021.120897
M.L. Zhang, Z.D. Zhang, Z.H. Zhao, et al., ACS Catal. 11(2021) 11103–11108.
doi: 10.1021/acscatal.1c02556
W.F. Xie, H. Li, G.Q. Cui, et al., Angew. Chem. Int. Ed. 60(2021) 7382–7388.
doi: 10.1002/anie.202014655
Q.N. Wang, C.Q. Zhu, C. Wu, H.B. Yu, Electrochim. Acta 319(2019) 138–147.
doi: 10.1016/j.electacta.2019.06.167
J. Wu, K. Zhu, H. Xu, W. Yan, Chin. J. Catal. 40(2019) 917–927.
doi: 10.1016/S1872-2067(19)63342-5
N. Nandal, S.L. Jain, Coordin. Chem. Rev. 451(2022) 214271.
doi: 10.1016/j.ccr.2021.214271
E. Kusmierek, Catalysts 10(2020) 439.
doi: 10.3390/catal10040439
S.S. Liu, Q.J. Xing, Y. Chen, et al., ACS Sustain. Chem. Eng. 7(2019) 1250–1259.
doi: 10.1021/acssuschemeng.8b04917
J. Zhang, S. Lv, J.L. Zheng, et al., ACS Sustain. Chem. Eng. 8(2020) 11133–11140.
doi: 10.1021/acssuschemeng.0c01906
J.F. de Brito, J.A.L. Perini, S. Perathoner, M.V.B. Zanoni, Electrochim. Acta 306(2019) 277–284.
doi: 10.1016/j.electacta.2019.03.134
D. Wang, Y.N. He, N. Zhong, et al., J. Hazard. Mater. 410(2021) 124563.
doi: 10.1016/j.jhazmat.2020.124563
D.B. Miklos, C. Remy, M. Jekel, et al., Water Res. 139(2018) 118–131.
doi: 10.1016/j.watres.2018.03.042
X.G. Duan, H.Q. Sun, S.B. Wang, Acc. Chem. Res. 51(2018) 678–687.
doi: 10.1021/acs.accounts.7b00535
J. Lee, U. von Gunten, J.H. Kim, Environ. Sci. Technol. 54(2020) 3064–3081.
doi: 10.1021/acs.est.9b07082
J.P. Zou, Y. Chen, S.S. Liu, et al., Water Res. 150(2019) 330–339.
doi: 10.1016/j.watres.2018.11.077
W.D. Oh, Z.L. Dong, T.T. Lim, Appl. Catal. B: Environ. 194(2016) 169–201.
doi: 10.1016/j.apcatb.2016.04.003
R. Cheula, M. Maestri, G. Mpourmpakis, ACS Catal. 10(2020) 6149–6158.
doi: 10.1021/acscatal.0c01005
H.Y. Tan, J. Wang, S.Z. Yu, K.B. Zhou, Environ. Sci. Technol. 49(2015) 8675–8682.
doi: 10.1021/acs.est.5b01264
M. Zhu, L.S. Zhang, S.S. Liu, et al., Chin. Chem. Lett. 31(2020) 1961–1965.
doi: 10.1016/j.cclet.2020.01.017
J. Wang, W. Liu, D.M. Li, Y.P. Wang, J. Alloy. Compd. 588(2014) 378–383.
doi: 10.1016/j.jallcom.2013.11.040
Z. Wang, C. Li, K. Domen, Chem. Soc. Rev. 48(2019) 2109–2125.
doi: 10.1039/C8CS00542G
L. Wang, X.L. Geng, L. Zhang, et al., Chemosphere 286(2022) 131558.
doi: 10.1016/j.chemosphere.2021.131558
C.G. Liu, Z.F. Lei, Y.N. Yang, Z.Y. Zhang, Water Res. 47(2013) 49586–49592.
X.J. Zheng, C.L. Li, M. Zhao, et al., Int. J. Hydrog. Energy 42(2017) 7917–7929.
doi: 10.1016/j.ijhydene.2016.12.131
Y. Rong, L. Tang, Y.H. Song, et al., RSC Adv. 6(2016) 80595–80603.
doi: 10.1039/C6RA15320H
K.H. Chu, L.Q. Ye, W. Wang, et al., Chemosphere 183(2016) 219–228.
L. He, L. Li, T.T. Wang, et al., Dalton Trans. 43(2014) 16981–16985.
doi: 10.1039/C4DT02557A
R. Shwetharani, M. Sakar, H.R. Chandan, R.G. Balakrishna, Mater. Lett. 218(2018) 262–265.
doi: 10.1016/j.matlet.2018.02.031
Y.P. Peng, H.L. Chen, C.P. Huang, Appl. Catal. B: Environ. 209(2017) 437–446.
doi: 10.1016/j.apcatb.2017.02.084
J. Han, Y.R. Bian, X.Z. Zheng, X.M. Sun, L.W. Zhang, Chin. Chem. Lett. 28(2017) 2239–2243.
doi: 10.1016/j.cclet.2017.08.031
A. Patsoura, D.I. Kondarides, X.E. Verykios, Appl. Catal. B: Environ. 64(2006) 171–179.
doi: 10.1016/j.apcatb.2005.11.015
J.G. Wang, P. Zhang, X. Li, J. Zhu, H.X. Li, Appl. Catal. B: Environ. 134-135(2013) 198–204.
doi: 10.1016/j.apcatb.2013.01.006
M.Q. Hu, Z.P. Xing, Y. Cao, et al., Appl. Catal. B: Environ. 226(2018) 499–508.
doi: 10.1016/j.apcatb.2017.12.069
K.X. Li, Z.X. Zeng, L.S. Yan, et al., Appl. Catal. B: Environ. 187(2016) 269–280.
doi: 10.1016/j.apcatb.2016.01.046
H. Park, A. Bak, Y.Y. Ahn, J. Choi, M.R. Hoffmannn, J. Hazard. Mater. 211-212(2012) 47–54.
doi: 10.1016/j.jhazmat.2011.05.009
Y.C. Nie, F. Yu, L.C. Wang, et al., Appl. Catal. B: Environ. 227(2018) 312–321.
doi: 10.1016/j.apcatb.2018.01.033
X.H. Jiang, L.C. Wang, F. Yu, et al., ACS Sustain. Chem. Eng. 6(2018) 12695–12705.
doi: 10.1021/acssuschemeng.8b01695
J.P. Zou, D.D. Wu, S.K. Bao, et al., ACS Appl. Mater. Interfaces 7(2015) 28429–28437.
doi: 10.1021/acsami.5b09255
D.K. Wang, H. Zeng, X. Xiong, et al., Sci. Bull. 65(2020) 113–122.
doi: 10.1016/j.scib.2019.10.015
F. Lu, D. Astruc, Coordin. Chem. Rev. 356(2018) 147–164.
doi: 10.1016/j.ccr.2017.11.003
K. Vikrant, K.H. Kim, Chem. Eng. J. 358(2019) 264–282.
doi: 10.1016/j.cej.2018.10.022
J.J. Rueda-Marquez, I. Levchuk, P.F. Ibanez, M. Sillanpaa, J. Clean. Prod. 258(2020) 120694.
doi: 10.1016/j.jclepro.2020.120694
S. Al-Amshawee, M.Y.B.M. Yunus, A.A.M. Azoddein, et al., Chem. Eng. J. 380(2020) 122231.
doi: 10.1016/j.cej.2019.122231
G.R. Xu, Z.H. An, K. Xu, et al., Coordin. Chem. Rev. 427(2021) 213554.
doi: 10.1016/j.ccr.2020.213554
Y. Zhu, W.H. Fan, T.T. Zhou, X.M. Li, Sci. Total Environ. 678(2019) 253–266.
doi: 10.1016/j.scitotenv.2019.04.416
Z. Xu, Q.R. Zhang, X.C. Li, X.F. Huang, Chem. Eng. J. 429(2022) 131688.
doi: 10.1016/j.cej.2021.131688
X. Zhao, L.B. Guo, B.F. Zhang, H.J. Liu, J.H. Qu, Environ. Sci. Technol. 47(2013) 4480–4488.
doi: 10.1021/es3046982
F. Zhang, W.L. Wang, C.Z. Zhou, Y.L. Sun, J.F. Niu, Chemosphere 278(2021) 130465.
doi: 10.1016/j.chemosphere.2021.130465
H.B. Zeng, S.S. Liu, B.Y. Chai, et al., Environ. Sci. Technol. 50(2016) 6459–6466.
doi: 10.1021/acs.est.6b00632
Z H, W.Q. Guo Wang, B.H. Liu, et al., Water Res. 160(2019) 405–414.
doi: 10.1016/j.watres.2019.05.059
D.K. Wang, H. Zeng, S.Q. Chen, J. Catal. 406(2022) 1–8.
doi: 10.1016/j.jcat.2021.12.027
L. Tian, P. Chen, X.H. Jiang, et al., Water Res. 209(2022) 117890.
doi: 10.1016/j.watres.2021.117890
S.C. Tian, C.Z. Dang, R. Mao, X. Zhao, ACS Sustain. Chem. Eng. 6(2018) 10273–10281.
doi: 10.1021/acssuschemeng.8b01634
K. Xiao, B. Zhou, S.Y. Chen, et al., Electrochem. Commun. 100(2019) 34–38.
doi: 10.1016/j.elecom.2019.01.018
X. Zhao, J.J. Zhang, M. Qiao, H.J. Liu, J.H. Qu, Environ. Sci. Technol. 49(2015) 4567–4574.
doi: 10.1021/es5062374
K.H. Xue, J. Wang, R. He, et al., Sci. Total Environ. 732(2020) 138963.
doi: 10.1016/j.scitotenv.2020.138963
X.H. Jiang, Q.J. Xing, X.B. Luo, et al., Appl. Catal. B: Environ. 228(2018) 29–38.
doi: 10.1016/j.apcatb.2018.01.062
J.M. Luo, S.Q. Zhang, M. Sun, et al., ACS Nano 13(2019) 9811–9840.
doi: 10.1021/acsnano.9b03649
Y.Y. Wu, Y.Q. Li, H.J. Hu, G.S. Zeng, C.H. Li, ACS ES & T Eng. 1(2021) 603–611.
X.X. Ma, X.K. Liu, J.H. Tang, et al., Appl. Surf. Sci. 602(2022) 154276.
doi: 10.1016/j.apsusc.2022.154276
M.S. Koo, X.F. Chen, K. Cho, T.C. An, W. Choi, Environ. Sci. Technol. 53(2019) 9926–9936.
doi: 10.1021/acs.est.9b02401
X.H. Jiang, L.S. Zhang, H.Y. Liu, et al., Angew. Chem. Int. Ed. 59(2020) 23112–23116.
doi: 10.1002/anie.202011495
L.S. Zhang, X.H. Jiang, Z.A. Zhong, et al., Angew. Chem. Int. Ed. 60(2021) 21751–21755.
doi: 10.1002/anie.202109488
M. Govindan, K.C. Pillai, B. Subramanian, I.S. Moon, ACS Omega 2(2017) 3562–3571.
doi: 10.1021/acsomega.7b00352
J. Kim, W. Choi, Energy Environ. Sci. 6(2010) 1042–1045.
J. Kim, D. Monllor-Satoca, W. Choi, Energy Environ. Sci. 5(2012) 7647–7656.
doi: 10.1039/c2ee21310a
G. Iervolino, V. Vaiano, J.J. Murcia, et al., J. Catal. 339(2016) 47–56.
doi: 10.1016/j.jcat.2016.03.032
D. Monllor-Satoca, R. Gómez, J. Phys. Chem. C 112(2008) 139–147.
doi: 10.1021/jp075672r
A. Heuer-Jungemann, N. Feliu, I. Bakaimi, et al., Chem. Rev. 119(2019) 4819–4880.
doi: 10.1021/acs.chemrev.8b00733
G. Hippargi, S. Anjankar, R.J. Krupadam, S.S. Rayalu, Fuel 291(2021) 120113.
doi: 10.1016/j.fuel.2020.120113
N. Sun, Y. Qu, C.H. Yang, et al., Appl. Catal. B: Environ. 263(2020) 118313.
doi: 10.1016/j.apcatb.2019.118313
Z.Y. Wu, Z.Y. Zhou, Y.J. Zhang, et al., Electrochim. Acta 254(2017) 140–147.
doi: 10.1016/j.electacta.2017.09.130
S.Q. Zhang, L.L. Wang, C.B. Liu, et al., Water Res. 121(2017) 11–19.
doi: 10.1016/j.watres.2017.05.013

Zhen Shi , Wei Jin , Yuhang Sun , Xu Li , Liang Mao , Xiaoyan Cai , Zaizhu Lou . Interface charge separation in Cu2CoSnS4/ZnIn2S4 heterojunction for boosting photocatalytic hydrogen production. Chinese Journal of Structural Chemistry, 2023, 42(12): 100201-100201. doi: 10.1016/j.cjsc.2023.100201
Shilong Li , Liang Duan , Qiusheng Gao , Hengliang Zhang . Reduction of methane emission from microbial fuel cells during sulfamethoxazole wastewater treatment. Chinese Chemical Letters, 2025, 36(6): 110997-. doi: 10.1016/j.cclet.2025.110997
Xingyan Liu , Chaogang Jia , Guangmei Jiang , Chenghua Zhang , Mingzuo Chen , Xiaofei Zhao , Xiaocheng Zhang , Min Fu , Siqi Li , Jie Wu , Yiming Jia , Youzhou He . Single-atom Pd anchored in the porphyrin-center of ultrathin 2D-MOFs as the active center to enhance photocatalytic hydrogen-evolution and NO-removal. Chinese Chemical Letters, 2024, 35(9): 109455-. doi: 10.1016/j.cclet.2023.109455
Jinshu Huang , Zhuochun Huang , Tengyu Liu , Yu Wen , Jili Yuan , Song Yang , Hu Li . Modulating single-atom Co and oxygen vacancy coupled motif for selective photodegradation of glyphosate wastewater to circumvent toxicant residue. Chinese Chemical Letters, 2025, 36(5): 110179-. doi: 10.1016/j.cclet.2024.110179
Jiqing Liu , Qi Dang , Liting Wang , Dejin Wang , Liang Tang . Applications of flexible electrochemical electrodes in wastewater treatment: A review. Chinese Chemical Letters, 2024, 35(8): 109277-. doi: 10.1016/j.cclet.2023.109277
Ziruo Zhou , Wenyu Guo , Tingyu Yang , Dandan Zheng , Yuanxing Fang , Xiahui Lin , Yidong Hou , Guigang Zhang , Sibo Wang . Defect and nanostructure engineering of polymeric carbon nitride for visible-light-driven CO2 reduction. Chinese Journal of Structural Chemistry, 2024, 43(3): 100245-100245. doi: 10.1016/j.cjsc.2024.100245
Yulong Liu , Haoran Lu , Tong Yang , Peng Cheng , Xu Han , Wenyan Liang . Catalytic applications of amorphous alloys in wastewater treatment: A review on mechanisms, recent trends, challenges and future directions. Chinese Chemical Letters, 2024, 35(10): 109492-. doi: 10.1016/j.cclet.2024.109492
Hongliang Zeng , Yuan Ji , Jinfeng Wen , Xu Li , Tingting Zheng , Qiu Jiang , Chuan Xia . Pt nanocluster-catalyzed hydrogen evolution reaction: Recent advances and future outlook. Chinese Chemical Letters, 2025, 36(3): 109686-. doi: 10.1016/j.cclet.2024.109686
Yaoyin Lou , Xiaoyang Jerry Huang , Kuang-Min Zhao , Mark J. Douthwaite , Tingting Fan , Fa Lu , Ouardia Akdim , Na Tian , Shigang Sun , Graham J. Hutchings . Stable core-shell Janus BiAg bimetallic catalyst for CO2 electrolysis into formate. Chinese Chemical Letters, 2025, 36(3): 110300-. doi: 10.1016/j.cclet.2024.110300
Yiming Fang , Huimin Gao , Kaiting Cheng , Liang Bai , Zhengtong Li , Yadong Zhao , Xingtao Xu . An overview of photothermal materials for solar-driven interfacial evaporation. Chinese Chemical Letters, 2025, 36(3): 109925-. doi: 10.1016/j.cclet.2024.109925
Jianan Hong , Chenyu Xu , Yan Liu , Changqi Li , Menglin Wang , Yanwei Zhang . Decoding the interfacial competition between hydrogen evolution and CO2 reduction via edge-active-site modulation in photothermal catalysis. Acta Physico-Chimica Sinica, 2025, 41(9): 100099-0. doi: 10.1016/j.actphy.2025.100099
Kun WANG , Wenrui LIU , Peng JIANG , Yuhang SONG , Lihua CHEN , Zhao DENG . Hierarchical hollow structured BiOBr-Pt catalysts for photocatalytic CO2 reduction. Chinese Journal of Inorganic Chemistry, 2024, 40(7): 1270-1278. doi: 10.11862/CJIC.20240037
Xuejiao Wang , Suiying Dong , Kezhen Qi , Vadim Popkov , Xianglin Xiang . Photocatalytic CO2 Reduction by Modified g-C3N4. Acta Physico-Chimica Sinica, 2024, 40(12): 2408005-. doi: 10.3866/PKU.WHXB202408005
Xianghai Song , Xiaoying Liu , Zhixiang Ren , Xiang Liu , Mei Wang , Yuanfeng Wu , Weiqiang Zhou , Zhi Zhu , Pengwei Huo . Insights into the greatly improved catalytic performance of N-doped BiOBr for CO2 photoreduction. Acta Physico-Chimica Sinica, 2025, 41(6): 100055-0. doi: 10.1016/j.actphy.2025.100055
Runhua Chen , Qiong Wu , Jingchen Luo , Xiaolong Zu , Shan Zhu , Yongfu Sun . 缺陷态二维超薄材料用于光/电催化CO2还原的基础与展望. Acta Physico-Chimica Sinica, 2025, 41(3): 2308052-. doi: 10.3866/PKU.WHXB202308052
Fangfang WANG , Jiaqi CHEN , Weiyin SUN . CuBi@Cu-MOF composite catalysts for electrocatalytic CO2 reduction to HCOOH. Chinese Journal of Inorganic Chemistry, 2025, 41(1): 97-104. doi: 10.11862/CJIC.20240350
Xue Dong , Xiaofu Sun , Shuaiqiang Jia , Shitao Han , Dawei Zhou , Ting Yao , Min Wang , Minghui Fang , Haihong Wu , Buxing Han . 碳修饰的铜催化剂实现安培级电流电化学还原CO2制C2+产物. Acta Physico-Chimica Sinica, 2025, 41(3): 2404012-. doi: 10.3866/PKU.WHXB202404012
Qin Li , Huihui Zhang , Huajun Gu , Yuanyuan Cui , Ruihua Gao , Wei-Lin Dai . In situ Growth of Cd0.5Zn0.5S Nanorods on Ti3C2 MXene Nanosheet for Efficient Visible-Light-Driven Photocatalytic Hydrogen Evolution. Acta Physico-Chimica Sinica, 2025, 41(4): 100031-. doi: 10.3866/PKU.WHXB202402016
Yuhao Guo , Na Li , Tingjiang Yan . Tandem catalysis for photoreduction of CO2 into multi-carbon fuels on atomically thin dual-metal phosphochalcogenides. Chinese Journal of Structural Chemistry, 2024, 43(7): 100320-100320. doi: 10.1016/j.cjsc.2024.100320
Xueyang Zhao , Bangwei Deng , Hongtao Xie , Yizhao Li , Qingqing Ye , Fan Dong . Recent process in developing advanced heterogeneous diatomic-site metal catalysts for electrochemical CO2 reduction. Chinese Chemical Letters, 2024, 35(7): 109139-. doi: 10.1016/j.cclet.2023.109139