
Dispersion and Filler Network Structure of Fibrillar Silicates in Elastomers
English
Dispersion and Filler Network Structure of Fibrillar Silicates in Elastomers
-
Key words:
- Fibrillar silicates
- / Dispersion
- / Filler network
- / Elastomers
-
INTRODUCTION
Elastomer (rubber) is of special high elasticity at room temperature, and thus it is an indispensable basic material, and has been widely used in aerospace, weaponry, and automobile tire, etc. A disadvantage of elastomer is that it is of low mechanical strength, and has to be reinforced by nanofillers for the industry application[1, 2]. Carbon black and nano-silicon dioxide are two most common traditional fillers and both exhibit good reinforcing effect on elastomers owing to the nanoscale dispersion[3]. In the past decades some newly developed nanofillers such as carbon nanotube[4-6] and graphene[2, 7, 8] are introduced into elastomer and show very promising reinforcing effect. However, the vast manufacture as well as nanoscale dispersion of these fillers is hard to achieve by a practically feasible method at a low cost.
Researchers in both academia and industry are looking for promising alternatives to take the place of these fillers in some applications. One of the substitutes is the natural nano-fiber, which is playing more and more important roles in reinforcement and cost reduction in polymer industry[9]. Fibrillar silicate (FS) is a kind of hydrated magnesium aluminum silicate mineral composed of many nano-fibrillar single crystals, such as attapulgite or palygorskite. The molecule formula of FS is Mg5[Al]Si8O20(HO)2(OH2)4·H2O. The constructing unit of FS is fibrillary nano-single crystal which is 0.1-3μm in length and 10-25 nm in diameter. These single crystals are compactly stacked into crystal bundles which furtherly aggregate into particles with diameters of 5-50μm[10-12]. The surface of FS is full of Si―OH or Al―OH[10], thus FS can be effectively modified with coupling agent such as organic titanate, silane coupling agent, organic ammonium salt or surfactants. Owing to its unique structure and low cost, FS is widely utilized as absorbent, catalyst and its carriers, densifying agent, adhesive agent, food additives, and nuclei agent for polymer or as a filler used to reduce cost[13, 14]. Meanwhile, FS has attracted massive interests in industry applications in recent years as a filler for reinforcing polymers[15, 16]. It is found that the interaction between FS single crystals is the weak Van de Walls force, thus FS can be easily separated and dissociated into single crystals in polar medium, such as water. Through conventional polymer melt blending, modified FS can easily dissociate into single crystals or crystal bundles with diameters less than 100 nm, and these single crystals or crystal bundles can uniformly disperse in elastomer matrix and greatly reinforce various kinds of elastomers such as styrene butadiene rubber (SBR), acrylonitrile butadiene rubber (NBR), ethylene-propylene ethylidene norbornene terpolymer (EPDM)[10, 17] etc. Because of the high elastic modulus and high heat resistance, FS/elastomer composites find applications in tire, conveyer belt and elastomer hose. By using conventional polymer melt blending, we have successfully prepared these FS/elastomer nano-composites with low cost and excellent properties. The interfacial interaction between FS and these elastomers has also been deeply studied.
It has been reported that the filler network structure plays an important role in the mechanical properties of the composites. Many previous studies have focused on the dispersion and network formation of traditional spherical fillers such as carbon black and silica as well as layered fillers[18]. However, to the best of our knowledge, the dispersion and network formation of FS in elastomers has not been reported yet. Therefore, in this study, the dispersion and network of FS in elastomer as well as their influencing factors were studied to provide guidance for the preparation of high performance FS/elastomer composites for its wide applications in industry.
EXPERIMENTAL
Surface Modification of FS and Preparation of Unvulcanized and Vulcanized Composites
Surface modification of FS: FS was dissolved in deionized water to obtain an aqueous solution with a concentration of 20 wt%-50 wt%. Then the solution was stirred at a speed of 800 r/min to allow FS to dissociate into single crystals. Then a mixture of modifier/alcohol with a concentration of 20% was added slowly into the solution, which was then treated for 30 min at 60℃. Different amounts of the modifier/alcohol mixture were used to study the influence of the amount of coupling agents. After that the solvent was removed, and then the obtained dry solid particles were grinded into powders.
The preparation of composite: Elastomers and FS were firstly melt-mixed by using a two-roll mill. The gap of the rolls was adjusted to the smallest value where the elastomer becomes fluidic. 40 phr of FS was added slowly into the melt to ensure good dispersion, followed by 10 phr of vulcanizer and crosslinking accelerator. The unvulcanized composite was put into a press vulcanizer and through compression molding the composite was obtained. The cross-linking time was determined by disc oscillating rheometer. The vulcanization temperature of SBR was 150℃, while that of NBR, SiR and EPDM was 160℃.
Materials
Styrene butadiene rubber (SBR1502) was provided by Jihua Synthesis Rubber Factory of China. Acrylonitrile butadiene rubber (NBR) shown in Table 1, was purchased from Nandi Company, Taiwan, China. Ethylene-propylene ethylidene norbornene terpolymer (EPDM, dual-edge J-4045) consisting of 53.0 wt%-59.0 wt% ethylene, having the third monomer of ethylidene norbornene (ENB), and having an iodine value of 19-25 was purchased from Jilin Petrochemical Industrial Co., Ltd. (China). Silicone rubber (SiR) was provided by Beijing Chemical 2nd Factory of China. Fibrillar silicate (325 mesh) was supplied by Dalian Global Mineral Company of China. Modifiers, g-glycidypropyltrimethoxy silane (C9H20O5Si, KH560), bis(3-triethoxysilylpropyl) tetrasulfide ([(C2H5O)3SiCH2CH2CH2]2S4, Si69) and g-aminopropyltriethoxy silane (NH2(CH2)3Si(OC2H5)3, KH550), were purchased from Shuguang Chemicals Company, Nanjing, China. The surface tension of all elastomers and modifiers used in this work are listed in Table 2 and Table 3, respectively. Dicumyl peroxide (DCP) and other chemical agents were purchased from chemical store.
Elastomer matrices NBR1051a NBR1051b NBR1051c NBR3365 NBR1965 Mooney viscosity (ML1+4 100℃) 65 42 25 65 65 Acrylonitrile content (%) 41 41 41 33 19 a, b, c contain 0 phr, 20 phr and 40 phr dioctyl phthalate (DOP) per 100 phr NBR respectively. Table 1. Elastomer materialsElastomers SBR EPDM NBR SiR Surface tension (N/m) 40 34.5 52.6* 20.4 *20℃, acrylonitrile content 27% Table 2. Surface tension for several elastomersSilane coupling agents KH560 Si69 KH550 FS Surface tension (N/m) 0.0494 0.024 0.0357 0.058 *20℃, on soda-lime glass determined Table 3. Surface tension for several silane coupling agentsCharacterization
A Hitachi H-800-1 Transmission Electronic Microscope (TEM) was used to observe the microtomed samples of the composites at-100℃. The acceleration voltage was 200 kV.
A CAMBRIGE S-250MK3 Scanning Electronic Microscope (SEM) was used to observe cryo-fractured surface morphology of the composites.
The Payne effect of FS/elastomer unvulcanized and vulcanized composites was investigated by a rubber process analyzer (RPA 2000, Monsanto Co., USA). The strain scanning conditions were 60℃, 1 Hz.
RESULTS AND DISCUSSION
Effect of Elastomer Polarity
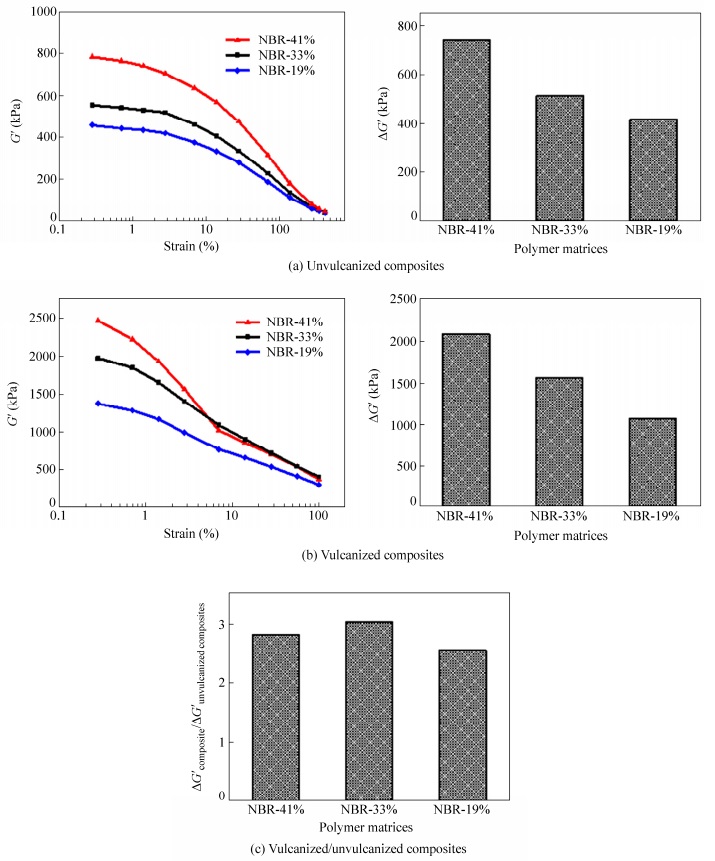
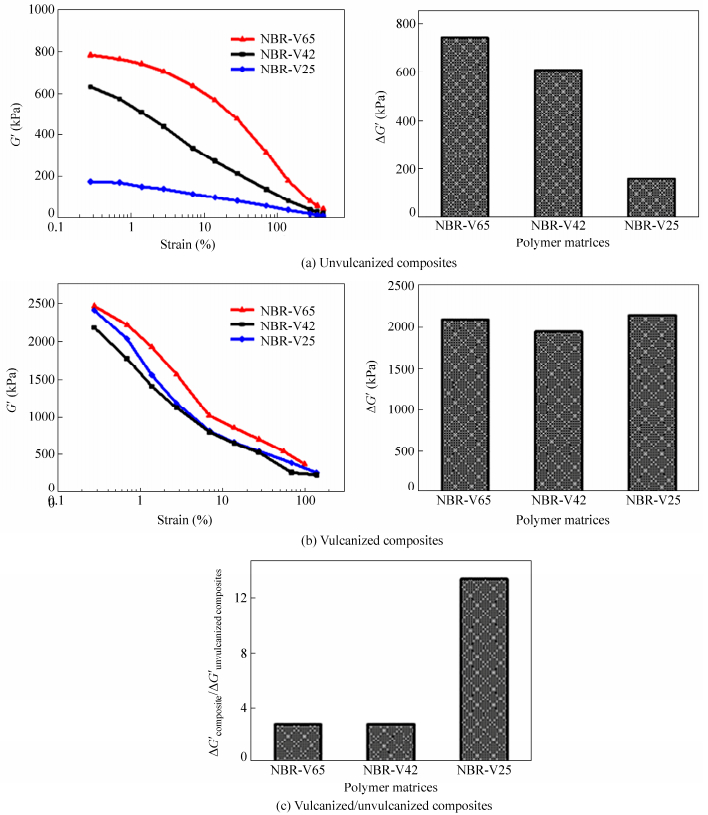
The content of acrylonitrile in NBR represents the molecular polarity of NBR. NBR with higher content of acrylonitrile shows higher molecular polarity. To determine the effect of matrix molecular polarity, NBR matrices with same Mooney viscosity and molecular weight but different contents of acrylonitrile are used to prepare FS/NBR composites. As shown in Fig. 5(a), FS/NBR unvulcanized composite with higher contents of acrylonitrile shows stronger Payne effect. Among these three unvulcanized composites, the filler network of FS/NBR-41% composite with the acrylonitrile content of 41% is the strongest, followed by FS/NBR-33% composite with the acrylonitrile content of 33%. The filler network of the FS/NBR-19% composite with the acrylonitrile content of 19% is the weakest. From Fig. 5(b), filler networks of these three NBR/FS vulcanized composites obey very similar rule to that of corresponding unvulcanized composites. However, the biggest change in filler network during vulcanization is found in FS/NBR-33% composite with the acrylonitrile content of 33% (see Fig. 5c), indicating that the re-aggregation of fillers is the strongest.
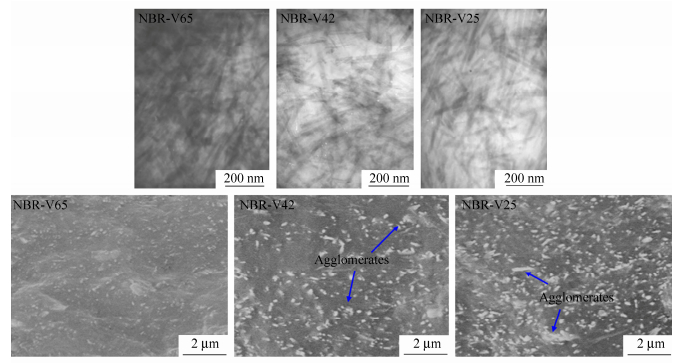
Figure 6 shows the SEM photos of cryo-fractured surfaces of the vulcanized composites. The dispersion of FS in NBR with the highest acrylonitrile content is the best. In the composites with low acrylonitrile contents there are a few FS agglomerates with diameters larger than 100 nm. Based on discussion above, take the effects of surface energy of filler and elastomer into consideration, we redress Eqs. (2) and (3) as Eqs. 2(a) and 3(a) respectively. Therefore, we can conclude that as the surface energy difference between filler and elastomer decreases, the force exerted on FS would increase to enhance the filler dispersion. Generally, NBR with high acrylonitrile content has higher surface energy than NBR with low acrylonitrile content, thus the surface energy difference between filler and NBR matrix with high acrylonitrile content is smaller and the filler dispersion is better. As discussed before, good dispersion of FS contributes to the formation of strong filler network. According to Eq. 3(a), the diffusion coefficient of filler increases as the surface energy difference between filler and elastomer decreases. Thus high molecule polarity may help to strengthen the FS re-aggregation. However, as shown in Fig. 5(c) the filler network of NBR/FS composite with 33% acrylonitrile content shows the strongest filler re-aggregation. This can be explained that the high molecule polarity also strengthens the interaction and entanglement of molecule chains, which is averse to FS diffusion. Thus the re-aggregation of fillers shows slightly different pattern with filler dispersion corresponding to acrylonitrile content.
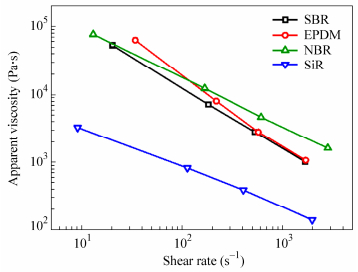
According to the influencing factors discussed above, the difference in filler dispersion and network formation in different elastomer matrices can be explained. Elastomers are usually pseudoplastic fluids (see Fig. 7), their apparent viscosity decreases as the shear rate increases. The surface tension of these elastomers is shown in Table 2. As a result, the difference in viscosity and surface tension of elastomer matrix leads to the difference in filler dispersion and network formation in composites. For FS/SiR composites, agglomerates of FS mainly disaggregate into smaller size through exfoliation during the shear process because of the low viscosity of SiR. In other words, the agglomerates observed in SiR composites stem from partial exfoliation during the blending process rather than re-aggregation during the vulcanization process. Thus, it can be concluded that uniform dispersion of FS would be hard to achieve in most plastics with low processing viscosity.
Dispersion and Filler Network of Fibrillar Silicates in Different Elastomers
As previously reported, the network of filler can be characterized by the Payne effect[18]. Composites with stronger filler network show stronger Payne effect. In other words, composites with stronger filler network show higher dynamic modulus (G’) at very small strains, their filler network breaks down fast as the exerted strain increases and their G’decreases remarkably once the filler network breaks down. For elastomer-filler system, dynamic modulus of the unvulcanized composite (unvulcanized blend) mainly depends on the filler network and the filler-elastomer bonding, while that of vulcanized composite depends on the combined effect of the filler network, the filler-elastomer bonding and elastomer network. This can be shown as the following Eq.(1).
The effect of pure elastomer’s network on dynamic modulus is so subtle compared with that of filler network that it can be neglected. FS is a kind of inorganic filler with strong polarity, the physical and chemical properties of which are quite similar to those of white carbon black. For unmodified FS, the FS-elastomer bonding is also weak enough that its effect can be neglected. Thus the dynamic modulus of either unvulcanized composite or vulcanized composite mainly depends on the filler network. Usually the modulus difference (ΔG’) between maximum and minimum of G’is used to characterize the filler network. That is composites with stronger filler network tend to show strongerΔG’. Additionally, the ration betweenΔG’of vulcanized composite and the counterpart of unvulcanized composite is used as an indication of the change in filler network during vulcanization.
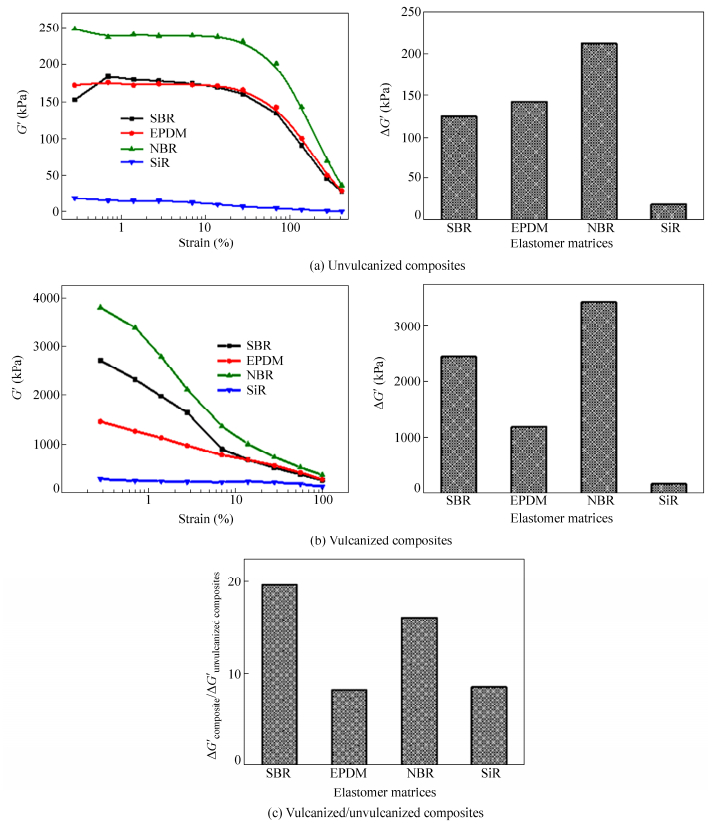
Different FS/elastomer unvulcanized composites and vulcanized composites are prepared to observe the difference in filler dispersion and network formation in different elastomer matrices. As shown in Fig. 1(a), the FS/SiR unvulcanized composite shows the lowest dynamic modulus and no obvious Payne effect was observed. For FS/NBR unvulcanized composite, the filler network is the strongest among these unvulcanized composites. Filler networks of unvulcanized composites based on matrices of nonpolar elastomers SBR and EPDM are very similar. From Fig. 1(b), we can see that the filler network of FS/NBR vulcanized composite is the strongest followed by that of FS/SBR vulcanized composite and FS/EPDM vulcanized composite, FS/SiR vulcanized composite shows no obvious Payne effect thus the filler network in which is very weak.
Figure 2 shows the TEM and SEM photos of FS/elastomer vulcanized composites with various elastomer matrices. In TEM pictures, light parts represent elastomer matrix and dark parts represent FS. From the cryo-fractured surface of FS/elastomer vulcanized composites it can be observed that FS are well dissociated into many fibrillary nano-single crystals and crystal bundles which are uniformly dispersed in the elastomer matrix. These nano-single crystals and crystal bundles overlap and interact to form filler network. Among these composites the best dispersion of FS is achieved in NBR matrix followed by SBR matrix. Agglomerates with large size can be observed in FS/EPDM composites, indicating a poorer dispersion than that of FS/NBR or FS/SBR composites. For FS/SiR composite, FS agglomerates with very large size are dispersed in the SiR matrix and no obvious filler network is observed, indicating that most of the FS are not exfoliated. It should be noted that TEM is not carried out for SiR composites because of the difficulty in ultrathin sectioning of SiR caused by the low glass transition temperature of SiR.
Effect of Silane Coupling Agent
Dispersion and Re-aggregation Mechanism of FS in Elastomer Matrix
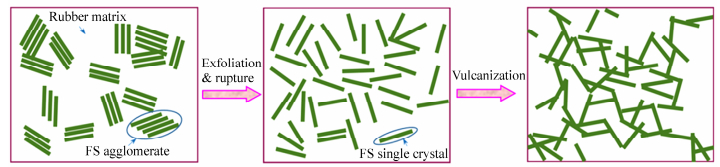
The dispersion of FS in elastomers is dominated by the simultaneous rupture and exfoliation process, different from that of spherical fillers. The scheme of dispersion and re-aggregation process of FS in elastomer matrix is shown in Fig. 11. When FS is added into an elastomer, FS agglomerates endure a shear force transferred from the matrix and the large scale agglomerates are exfoliated into submicron particles (< 1μm) owing to the low interaction force. Under further exfoliation, these submicron particles can be dissociated into nano-single crystals and crystal bundles. This is quite different from silica because silica exists in elastomers as spherical particles or particle agglomerates, which usually need to be modified to obtain a good dispersion because of the poor compatibility between silica and elastomer matrix. And as FS nano-single crystals and crystal bundles are dispersed uniformly in elastomer matrix, the fibrillary crystals partly overlap and thus interact to achieve a local network. Under limited volume fraction of FS, good dispersion may increase the possibility of the intertwining of FS to form a universal strong filler network. During vulcanization, these nano-single crystals and crystal bundles absorb energy that stems from high temperature and tend to re-aggregate. However, the energy absorbed is not enough for the nano-single crystals and crystal bundles to re-crystalize into the original large agglomerates. On the other hand, during the vulcanization process, the high temperature can result in the decrease in viscosity of the elastomer matrix, facilitating the re-aggregation of FS. Although the uniformity of the dispersion of FS is disrupted, the nano-single crystals and crystal bundles are able to overlap and interact with one another. Through overlapping and intertwining, a stronger filler network is formed.
Studies have proven that particles with nano-dispersion may re-aggregate when the elastomer matrix is in rubbery state or viscous state during processing. The re-aggregation can result in a universal stereo network of fillers which can lead to high modulus in both vulcanized composite and unvulcanized composite. Excessive re-aggregation may lead to a transition from universal stereo network to local stereo network and finally densely-packed agglomerates. On one hand, re-aggregation is controlled thermodynamically: the large difference in surface energy between filler and matrix, high volume fraction of filler, small particle size and large shape coefficient favor the filler re-aggregation. On the other hand, re-aggregation is also controlled kinetically: the low matrix viscosity and small ligament between fillers would contribute to filler re-aggregation. FS in matrix like NBR form the network structure because of the following three reasons. First, the surface energy of FS is lower than traditional fillers thus the physical motivation of filler re-aggregation is low. Second, FS is very easy to dissociated into single crystals and the high viscosity of matrix and large ligament between fillers further prevent the re-aggregation of the fillers during vulcanization, favoring the achievement of a good dispersion of FS. Third, the FS single crystals are rather easy to form network owing to the large aspect ratio. Thus, FS may form a network structure rather than large aggregates.
Effect of Elastomer Viscosity
Among composites with various elastomer matrix, the FS/NBR composite exhibits the strongest filler network and thus NBR was chosen as the matrix to evaluate the influence of others factors on the filler dispersion and network structure. Various amounts of extended oil DOP were used to prepare NBR matrix with different Mooney viscosity (see Table 1). As shown in Fig. 3(a), at the same volume fraction of FS, the dynamic modulus decreases faster and the filler network is stronger for the unvulcanized NBR composites with higher matrix Mooney viscosities. Especially, the filler network of the unvulcanized composite is very weak when the Mooney viscosity of NBR matrix is very low. According to Eq.(2), during simple shear process, the maximum force exerted on the filler agglomerates is proportional to the shear viscosity of matrix, shear rate and shape coefficient of filler[19].
For ellipsoidal agglomerates with the larger ratios of macro axis and short-axis:
where Fh, max: maximum shear; ηm: shear viscosity; γ: shear rate; L: the length of ellipsoidal agglomerate; ra: the macro axis/short-axis ratio of ellipsoidal agglomerate.
According to Eq. (2), the force exerted on FS during shear blending process in NBR matrix with high Mooney viscosity is stronger than that in NBR matrix with low Mooney viscosity. As a result, the agglomerates of FS are more easily to dissociate into single crystals or crystal bundles of small size in NBR matrix with higher Mooney viscosity, which facilitates the dispersion of FS. For traditional spherical fillers, good dispersion in elastomer matrix will lead to weakened filler network because of the decrease of filler interaction. However, as the data suggest, good dispersion of FS in elastomer matrix favors the formation of a strong filler network. This can be ascribed to the overlapping of fibrillary structure which strengthens the interaction of fillers. From Fig. 3(b), after vulcanization, the filler network of the vulcanized composite is much stronger than that of the unvulcanized composite. It can be explained that the heat history accelerates the re-aggregation of filler and strengthens the filler network during the vulcanization process. Therefore, the dynamic moduli of the vulcanized composites increase dramatically[20]. It is also found that Payne effects of the three FS/NBR vulcanized composites are close to each other. As shown in Fig. 3(c), the change in filler network after vulcanization is remarkable for composites with low Mooney viscosity matrix. This implies that filler network formed through filler re-aggregation is stronger in NBR matrix with lower Mooney viscosities. This is because the filler re-aggregation predominately depends on the filler diffusion coefficient, which decreases with the increase in viscosity of the matrix according to Eq. (3)[20]. FS in NBR matrix with lower viscosity endure a stronger re-aggregation during vulcanization process.
whereΔ: diffusion coefficient of medium; K: Boltzmann constant; T: temperature; η: viscosity of matrix; a: radius of particle.
Based on the above results, the dispersion and network formation of FS in vulcanized composite is determined by both dispersing process and re-aggregating process. High viscosity of matrix is beneficial to the dispersion of FS, but prevents FS from re-aggregating. Thus, FS/NBR composites with different NBR Mooney viscosities show little difference in Payne effect. The final filler network of FS/NBR vulcanized composite with higher viscosity is slightly stronger.
TEM and SEM pictures of FS/NBR vulcanized composites with different NBR Mooney viscosities are shown in Fig. 4. It can be seen from TEM pictures that most FS in all the composites are dissociated and dispersed uniformly as single crystals or crystal bundles with diameters smaller than 100 nm. From SEM pictures, it is observed that a few agglomerates with diameters of 100-500 nm still exist. However, they are much smaller compared with the original size of FS agglomerates. In addition, it can be concluded that NBR with higher Mooney viscosity is conducive to achieve good dispersion of FS, and again it is proven that good dispersion of FS contributes to the formation of strong filler network, which is coherent with former discussion.
Types of silane coupling agent
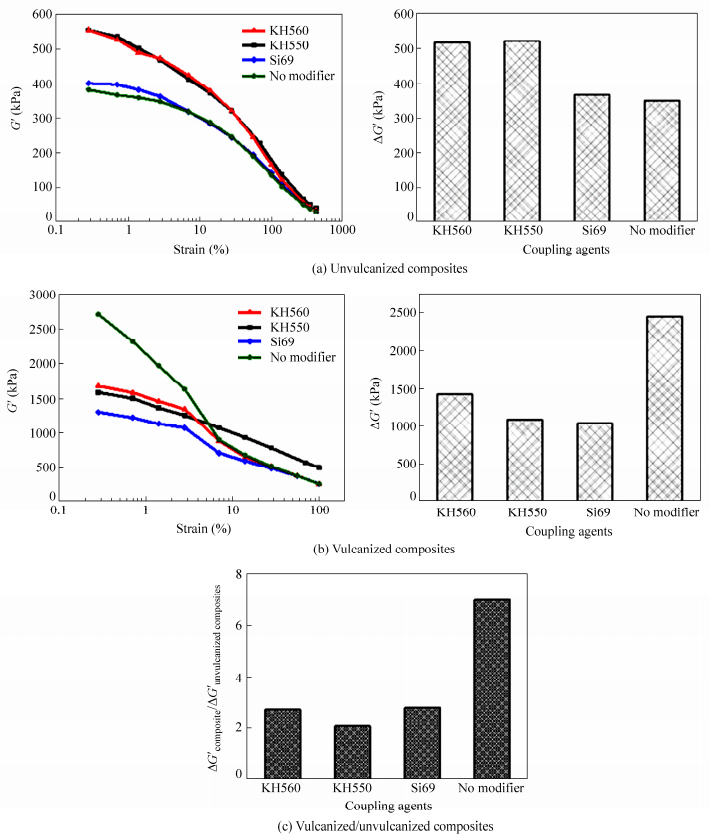
Silane coupling agent is widely used in surface modification of inorganic fillers to enhance the compatibility between filler and elastomer so as to improve the filler dispersion and the filler-elastomer bonding. As discussed above, the dissociation and dispersion of FS in polar matrix like NBR are better than those in nonpolar matrix like SBR and EPDM, especially in SiR matrix. Therefore, nonpolar elastomer SBR was chosen as the matrix to study the effect of the type and the loading of silane coupling agent on the dispersion and network formation of FS in elastomers. As shown in Fig. 8(a), the Payne effects of modified FS/SBR unvulcanized composites with different types of coupling agents are different. The Payne effects of KH550 or KH560 modified FS/SBR unvulcanized composites are the strongest, indicating that the filler networks in these two unvulcanized composites are the strongest. The network of Si69 modified FS is much weaker, and that of unmodified FS is the weakest (although close to that of Si69 modified FS). As discussed before, good dispersion of FS leads to strong filler network, thus the results indicate that the dispersion of KH550 or KH560 modified FS is better than that of Si69 modified FS and unmodified FS in SBR matrix. As shown in Fig. 8(b), the filler network of unmodified FS/SBR composite largely increases after vulcanization and it is stronger than that of modified FS/SBR vulcanized composites. For the modified FS/SBR vulcanized composites, the filler network of KH560 modified FS is the strongest; followed by that of KH550 modified FS and that of Si69 modified FS. As shown in Fig. 8(c), the re-aggregation of unmodified FS during vulcanization is much stronger than that of modified FS.
As shown in Fig. 9, many agglomerates with diameters 100-400 nm can be observed from the cryo-fractured surface of unmodified FS/SBR vulcanized composites. The dispersion of modified FS is much better than that of unmodified FS, which is coherent with the former discussion that good dispersion of FS contributes to strong filler network. Among the three modified FS/SBR composites, the dispersion of KH550 modified FS is the best, many fibrillary single crystals or crystal bundles can be observed in the composite, followed by the dispersion of KH560 modified FS and that of Si69 modified FS. This again proves that the final dispersion of FS in composite is determined by both the dispersing process and the re-aggregating process.
In fact, the results above attribute to the modification mechanism of different silane coupling agents. First, the silane coupling agent is adsorbed on the surface of FS single crystal, then the alkoxyls at one end of the silane coupling agent hydrolyze into silanols, which condensate with hydroxyls of FS to form Si―O―Si bonds[21]. In this process, the organic modifier can weaken the interaction between single crystals and lower the surface tension of FS thus enhance its compatibility with elastomer. This is beneficial to the dissociation of FS under shear during the elastomer melt blending process and can prevent single crystal from re-agglomerating into large particles, so the dispersion of FS in elastomer is improved. Different silane coupling agents show different effects on the dispersion of modified FS. The surface tensions of KH560 (silane coupling agent with a polar epoxy group) and KH550 (silane coupling agent with a polar aminopropyl group) are closer to the surface tension of FS (See Table 3) and are higher than that of Si69 (with no polar group). These two coupling agents, by which FS can be well wetted, can be easily adsorbed and fastened onto FS surface by Si―O―Si bond[21]. Consequently, under the same loading the dispersion of KH560 or KH550 modified FS is better than that of Si69 modified FS. On the other hand, the long organic chains or organic functional groups at the other end of silane coupling agent can tangle or react with macromolecules, which can improve the interfacial adhesion between filler and elastomer. For modified FS/SBR composites, if the vulcanizer is sulfur, aminopropyl groups in KH550, epoxy groups in KH560 and polysulfur groups in Si69 may participate in the vulcanization reaction, which will improve the filler-elastomer bonding and thus prevent FS from re-aggregating. As a result, the filler re-aggregation of modified FS is weaker than unmodified FS.
The amount of silane coupling agent
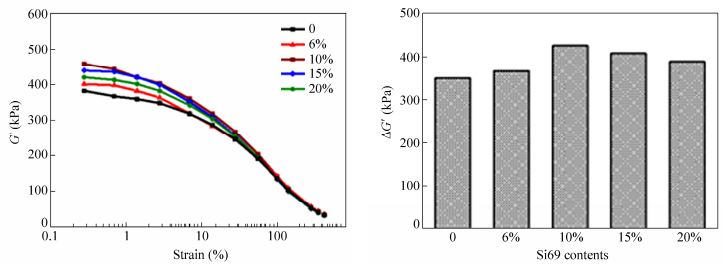
Si69 was chosen to evaluate the influence of the amount of silane coupling agent on the dispersion and network formation of FS in elastomers. As shown in Fig. 10, the Payne effects of Si69 modified FS/SBR unvulcanized composites are much stronger than that of the unmodified FS/SBR unvulcanized composite. As the loading of Si69 increases, the resulting filler network of the unvulcanized composite becomes stronger. The strongest filler network is achieved when the loading of Si69 is 10% of the weight of FS, above that loading any increase in Si69 loading will result in a weakened filler network. That is because during surface modification process, the excessive Si69 adsorbed on single crystals or crystal bundles of FS may form multi-layer adsorption, which is averse to the filler network formation. On the other hand, the functional groups of FS are limited thus excessive Si69 cannot contribute to the separation of extra FS bundles to increase the amount of filler network constructing units (single crystals). So excessive Si69 will weaken the bonding between single crystals and thus weaken the filler network, besides it can also increase the cost of the composite.
CONCLUSIONS
The dispersion of FS directly influences the filler network of the unvulcanized composite. Unlike traditional spherical fillers, FS with good dispersion in elastomer matrix during mechanical blending contributes to a strong FS filler network owing to the overlapping of FS. Vulcanization of elastomers greatly strengthens the filler network owing to the filler re-aggregation. The final dispersion and filler network of FS in composites is determined by the combined effect of dispersion and the re-aggregation of FS. The results showed that high viscosity of elastomer matrix contributes to good dispersion and strong filler network. High molecular polarity contributes to good dispersion of FS in unvulcanized composites. In vulcanized composites, the filler re-aggregation during vulcanization increases with the increase in molecular polarity, but when the molecular polarity reaches a certain point, the re-aggregation is prevented. The modification of silane coupling agent contributes to the good dispersion of FS in unvulcanized composites, and can prevent filler re-aggregation. Among KH550, KH560 and Si69, KH560 shows the best modification effect in FS/SBR composites. Within a certain amount, the increase of the amount of silane coupling agent results in strengthened filler network, while excessive silane coupling agent weakens the filler network.
-
-
[1]
Podsiadlo, P., Kaushik, A.K., Arruda, E.M., Waas, A.M., Shim, B.S., Xu, J., Nandivada, H., Pumplin, B.G., Lahann, J. and Ramamoorthy, A., Science, 2007, 318(5847): 80
-
[2]
Ramanathan, T., Abdala, A., Stankovich, S., Dikin, D., Herrera-Alonso, M., Piner, R., Adamson, D., Schniepp, H., Chen, X. and Ruoff, R., Nature Nanotechnol., 2008, 3(6): 327
-
[3]
Zou, H., Wu, S. and Shen, J., Chem. Rev, 2008, 108(9): 3893
-
[4]
Lorenz, H., Fritzsche, J., Das, A., Stöckelhuber, K., Jurk, R., Heinrich, G. and Klüppel, M., Compos. Sci. Technol., 2009, 69(13): 2135
-
[5]
Noguchi, T., Magario, A., Fukazawa, S., Shimizu, S., Beppu, J. and Seki, M., Mater. Trans., 2004, 45(2): 602
-
[6]
Spitalsky, Z., Tasis, D., Papagelis, K. and Galiotis, C., Prog. Polym. Sci., 2010, 35(3): 357
-
[7]
Ozbas, B., O'Neill, C.D., Register, R.A., Aksay, I.A., Prud'homme, R.K. and Adamson, D.H., J. Polym. Sci., Part B: Polym. Phys., 2012, 50(13): 910
-
[8]
Chen, Z. and Lu, H., J. Mater. Chem., 2012, 22(25): 12479
-
[9]
Liu, L., Qi, Z. and Zhu, X., J. Appl. Polym. Sci., 1999, 71(7): 1133
-
[10]
Tian, M., Liang, W., Rao, G., Zhang, L. and Guo, C., Compos. Sci. Technol., 2005, 65(7): 1129
-
[11]
Wang, L. and Sheng, J., Polymer, 2005, 46(16): 6243
-
[12]
Murray, H.H., Appl. Clay Sci., 2000, 17(5): 207
-
[13]
Yan, S., "Clay miner", Beijing Publisher, Beijing, 1981
-
[14]
Xu, W. and He, P., J. Appl. Polym. Sci., 2001, 80(2): 304
-
[15]
Gao, J., Zhang, Q., Wang, K., Fu, Q., Chen, Y., Chen, H., Huang, H. and Rego, J.M., Composites Part A: Appl. Sci. Manufact., 2012, 43(4): 562
-
[16]
Tian, M., Qu, C., Feng, Y. and Zhang, L., J. Mater. Sci., 2003, 38(24): 4917
-
[17]
Tian, M., Zhang, X., Zhang, L., Yin, S., Nishi, T. and Ning, N., Compos. Sci. Technol., 2013, 79: 21
-
[18]
Payne, A.R., J. Appl. Polym. Sci., 1962, 6(19): 57
-
[19]
Jeffery, G.B., "In proceedings of the royal society of london a: mathematical, physical and engineering sciencesed.", Vol. 102, The Royal Society, 1922, p.161
-
[20]
Lin, C.J., Hergenrother, W., Alexanian, E. and Bohm, G., Rubber Chem. Technol., 2002, 75(5): 865
-
[21]
Caravajal, G.S., Leyden, D.E., Quinting, G.R. and Maciel, G.E., Anal. Chem., 1988, 60(17): 1776
-
[1]
-

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 0
- 文章访问数: 1818
- HTML全文浏览量: 31




 下载:
下载:

