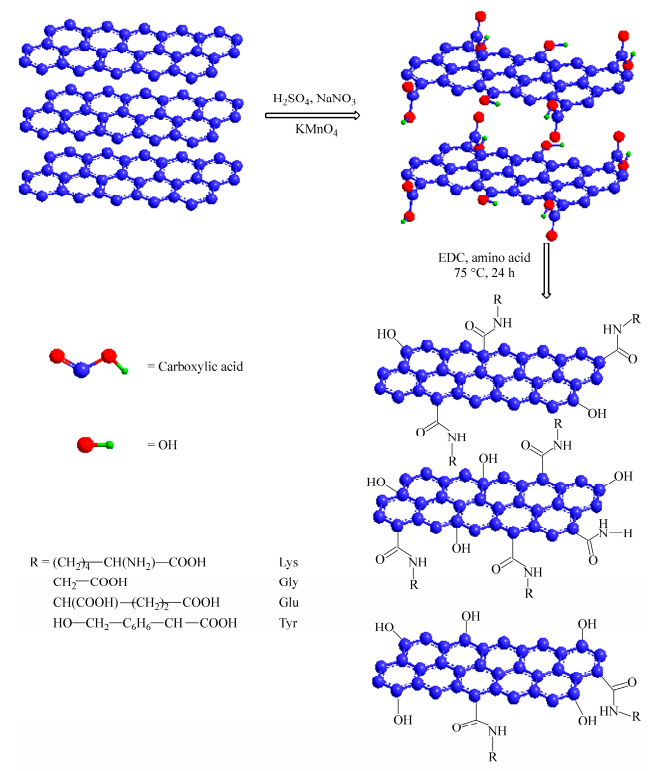
 Figure 1.
Schematic of the GO functionalization with different types of amino acids
Figure 1.
Schematic of the GO functionalization with different types of amino acids

Amino Acids Functionalized Graphene Oxide for Enhanced Hydrophilicity and Antifouling Property of Poly(vinylidene fluoride) Membranes
English
Amino Acids Functionalized Graphene Oxide for Enhanced Hydrophilicity and Antifouling Property of Poly(vinylidene fluoride) Membranes
-
Key words:
- PVDF membrane
- / Graphene oxide
- / Amino acids
- / Hydrophilicity
- / Antifouling property
-
INTRODUCTION
As a membrane material, polymeric material poly(vinylidene fluoride) (PVDF) has been received a great deal of attention and is commonly used in microfiltration membrane fields because of its excellent chemical resistance, good thermal stability, easily-controlled porosity and and outstanding mechanical properties[1-5]. However, the application of PVDF in water treatment field is restricted due to its low surface energy and hydrophobicity. In addition, the higher hydrophobicity of the PVDF membrane can result in membrane fouling by biomolecules and other organic matter, which limits its application in the fields of biotechnology and water treatment[6, 7]. Accordingly, developing a membrane with excellent surface hydrophilicity and outstanding antifouling (anti-organic fouling and anti-biofouling) performance is very vital for the membrane's practical application. Common methods of improving the hydrophilicity and antifouling property of PVDF membranes generally include surface modification[8, 9] and blending modification. Surface modification, especially grafting or adsorbing of hydrophilic polymers, can improve the hydrophilicity and pollution resistance ability of membranes[10, 11]. Yuan[12] had tethered a glycopolymer poly(d-gluconamidoethyl methacrylate) (PGAMA) onto the membrane surface via activators generated by electron transfer atom transfer radical polymerization (AGET ATRP) directly initiated from the PVDF surface. In Sun’s paper[13], surface-initiated atom transfer radical polymerization was used to tether poly(2-dimethylaminoethyl methacrylate) (PDMAEMA) onto microporous PVDF membranes in order to synthesize membrane adsorbers for protein adsorption.
Another method to improve the hydrophilicity of PVDF membranes is blending copolymers as additives (e.g. siloxane[14] and hydrophilic carbon materials such as carbon nanotubes[15]) with the bulk membrane materials during membrane preparation[16, 17]. This type of copolymer possesses good compatibility with the PVDF matrix and imparts antifouling resistance throughout the membrane. When blended with the polymer solution, the additives can migrate to the membrane surface in a water-based coagulation bath, which can create a hydrophilic surface.
Recently, graphene derivates have also become ideal candidates for polymer reinforcement on account of their unique architecture and superior performance. Correspondingly, it can be incorporated into microfiltration membranes to optimize targeted properties through the solution-blending method, such as high water permeability, better mechanical and antifouling performance[18-20].
Graphene oxide (GO) is an oxygen-containing graphene derivative with partial breakage of sp2-sp2 bonds into sp3-sp3 bonds for inserting some pendent groups like hydroxy, epoxy, and carboxylic acid on the surface[21], which can provide the reactive site for covalent functionalization. Graphene oxide derivatives functionalized with peptides, proteins, aptamers, avidin-biotin and other small biomolecules usually are used as building blocks. Chemical functionality of GO significantly alters the Vander Waals interaction among nanofiller aggregates, making them easy to be dispersed in the polymer matrix. Moreover, the functionalized GO can tightly intertwine with PVDF matrix. As a result, the covalent functionalization not only makes the dispersibility of GO better but also enables the interfacial interaction between graphene oxide and matrix to be stronger[22, 23].
Polyzwitterionic materials are intriguing compounds that have been well demonstrated for their super low-fouling ability to resist nonspecific protein adsorption and cell adhesion[24-27]. They can be further classified into betaines carrying a positive and a negative charge on the same unit, and polyampholytes carrying 1:1 positive and negative charge on two different monomer units such as mixed charge complex and natural amino acids (Glu-, Asp-, Lys+, and Arg+)[28, 29].
Similar to cysteine, lysine (abbreviated as Lys) contains two amine groups (―NH2) and one carboxyl group (―COOH), which is expected to be a new strategy for anti-fouling material design. At neutral pH, amino group and carboxyl group are protonated and deprotonated, respectively. When one amino group is consumed, the remained amino group and carboxyl group forms anion-cation ion pair and could be regarded as zwitterions. Previously, lysine was chemically grafted onto the membranes surface and lysine modified membranes showed superior protein fouling resistance under both static exposure and dynamic filtration conditions[30].
Hence, we can expect that adding the amino acids functionalized graphene oxide into membranes will enhance hydrophilicity and antifouling performance of the hybrid ultrafiltration membrane greatly. The present paper addresses the above-mentioned issue by introducing chemically functionalized GO, which was modified by amino acids (lysine, glycine, glutamic acid and tyrosine). Subsequently, the lysine functionalized GO (GO-lysine), which possessed better dispersion stability, is incorporated into PVDF matrix via phase inversion induced by the immersion precipitation technique to prepare the PVDF/GO-lysine membranes. Lysine has a wealth of hydrophilic polymer chains, which can penetrate into and entangle with PVDF matrix. As a result, a significant improvement of membrane antifouling property and hydrophilicity performance was achieved. Importantly, the targeted application of the membranes we made was to make antifouling membranes for water treatment and blood purification.
EXPERIMENTAL
Antifouling Property of the PVDF/GO-lysine Membranes
Materials
Natural graphite powder (diameter 5-10 μm, thickness 4-20 nm, layers < 30 and purity > 99.5 wt%), was purchased from Qingdao Tianhe Graphite Co. Ltd. (Shandong, China). Concentrated sulfuric acid (H2SO4, 98 wt%), hydrochloric acid (HCl), potassium permanganate (KMnO4), hydrogen peroxide aqueous solution (H2O2, 30 wt%), absolute ethanol and sodium nitrate (NaNO3), 1-ethyl-(3-3-dimethylaminopropyl)-carbodiimide hydrochloride (EDC), lysine, glycine, glutamic acid, tyrosine and polyvinylpyrrolidone (PVP) were purchased from Sinopharm Chemical Reagent Co., Ltd. PVDF (FR-904) (Mn = 3.8 × 105 g/mol) was obtained from Shanghai 3F new materials Co. LTD and was dried at 80 ℃ for 24 h before using. N-methyl-2-pyrrolidone (NMP) and other reagents were all chemically purified and were used directly. The deionized water was used throughout the experiments.
Hydrophilicity of PVDF/GO-lysine Membranes
To characterize the hydrophilicity of the membranes, contact angle (CA) of membranes was recorded at room temperature by a water contact angle system (OCA20, Dataphysics, Germany). Five different points of every sample were measured and the CA value was the average of these measurements. The solid-liquid interfacial free energy of hydration of membranes was also determined by the following Young-Dupré equation[32]:
where θ is the average contact angle and γwTOT (= 72.8 mJ•m-2 for pure water at 298.15 K) is the liquid surface tension. The free energies of hydration (ΔGSW) of membranes with water were calculated in order to quantify their relative “hydrophilicity”[33].
Preparation of Amino Acids Functionalized GO (GO-amino acids)
Amino acids were chosen to fabricate functionalized graphene oxide. At first, GO powder (100 mg) was dispersed in deionized water (50 mL) and then treated with ultrasonic oscillation for 2 h. Then an moderate amount of DCC and different amounts of amino acids (lysine, glycine, glutamic acid and tyrosine) were added into the GO solution, respectively. The mixture was under magnetic stirring for 24 h at 75 ℃. After completion the reaction, the mixture was centrifuged at 8000g-force using a Sorvall RC 6+ ultracentrifuge (Thermo Scientific, Marietta, OH) and the resulting precipitate was washed well with H2O/EtOH. Finally, the GO-amino acids were obtained with the precipitate dried at 60 ℃. And the schematic of the GO functionalized with different types of amino acids is shown in Fig. 1.
Preparation of PVDF/GO-lysine Membranes
The PVDF/GO-lysine membranes were fabricated by the classical phase inversion method and the specific formula was shown in Table 1. After the GO-lysine was added into NMP solvent and sonicated for 2 h (200 Hz), PVP and PVDF powders were added. Then the casting solution was magnetic stirred at 70 ℃ for 24 h. After fully degassed, the casting solution was spread onto a clean glass plate with a 150 μm gap and then the glass plate was immersed into a 60 ℃ coagulation bath (distilled water) for 15 min. Peeled off from the glass plate, the resultant membrane was rinsed with distilled water thoroughly. In order to identify these membranes easily, it was denoted as PVDF (M0), and PVDF/GO-lysine-2% (M1), PVDF/GO-lysine-4% (M2), PVDF/GO-lysine-6% (M3) and PVDF/GO-lysine-8% (M4), respectively.
Membrane ID PVDF GO-lysine PVP NMP M0 19 0 6 75 M1 17 2 6 75 M2 15 4 6 75 M3 13 6 6 75 M4 11 8 6 75 Characterization of GO and Amino Acid-GO
The presence of amino acid on the GO nanosheet was investigated by Fourier-transform infrared spectroscopy (FTIR, Thermo Nicolet 360, USA) at 4 cm-1 resolution and scanned at wavenumber range 400-4000 cm-1. X-ray diffraction (XRD) was used to characterize the crystalline structure of the composites. XRD patterns were collected using a Bruker, D8 Advanced diffractometer with a copper target at the wave length of λ CuKα = 0.1542 nm and a tube voltage of 40 kV and tube current of 35 mA, in the range of 5°-50° at the speed of 0.05 (°)/min. Morphology and structure of the GO-amino acids were observed with transmission electron microscopy (TEM) (TECNAI G2F20, FEI Co., USA) at voltage of 150 kV. Zeta potential was performed using a laser scattering method (ZETASIZER, 3000). Thermogravimetric analysis (TGA) was performed with a Setaram Setsys 1750 TG/DTA instrument at the heating rate of 5 K/min from 0 ℃ to 800 ℃ under N2 atmosphere.
In view of the zeta potential, dispersion stability and TGA results, the GO-lysine was chosen as the additive to further modificate the virgin PVDF membrane.
Characterization of PVDF/GO-lysine Membranes
The surface chemical composition of the membranes was analyzed by X-ray photoelectron spectroscopy (XPS) (PHI 5000C ESCA system, PHI Co., USA). The survey spectra was collected at a fixed analyzer pass energy of 160 eV. XPS analysis was conducted on the side of the membrane facing the water bath during the precipitation step of fabrication.
Membrane microstructures were observed under a field emission microscope (SEM, SIRION-100, FEI Co. Ltd). All specimens were coated with a thin layer of gold before observation.
The surface zeta potential was determinated via the zeta potential planar cell along with tracer particles (Zetasizer Nano instrument, Malvern). In principle, a high zeta potential (either positive or negative) should provide sufficient repulsive energy to the particles to maintain particle distance and protect sample stability by preventing flocculation and aggregation. The reported values are the average of the zeta potentials determined in two opposite flow directions and with at least three samples for each type of membranes.
Synthesis of Graphene Oxide
The GO was synthesized by the Hummer’s method with some modification[31]. The specific steps for the synthesis were as follows: First, a certain volume of concentrated H2SO4 (69 mL) was added to the mixture of graphite flakes (3.0 g) and NaNO3 (1.5 g) followed by cooling to 0 ℃ with an ice bath. Then, 9.0 g KMnO4 was added to the mixture slowly. And the mixture was then warmed to 35 ℃ and magnetic stirred for 30 min. An amount of additional water (138 mL) was added slowly with magnetic stirring for another 30 min. After completing the reaction, 5 mL 30% H2O2 was added to the mixture. And bubbles from the aqueous mixture and a color change to brilliant yellow were observed. After settling for approximately 12 h, the clear supernatant of the mixture was decanted. The precipitate was washed repeatedly with distilled water, and then centrifuged at 8000g-force using a Sorvall RC 6+ ultracentrifuge (Thermo Scientific, Marietta, OH). The sediment remaining at the bottom of the centrifuge tube went through at least three cycles of resuspension in DI water and then centrifuged to completely wash out chemical residuals. The pure GO powders were obtained by the freeze drying treatment for sediment.
Dynamic antifouling performance
The permeation flux and rejection of membranes were measured by dynamic ultrafiltration experimental equipments. The antifouling properties under dynamic flow conditions were evaluated through protein ultrafiltration experiments at ambient temperature. A dead-end stirred cell (Model 8200, Millipore Co., USA) with the effective membrane area 5.0 cm2 was used for the ultrafiltration study. Measuring steps were depicted as follows: each membrane was initially compacted with deionized water for 30 min at 0.2 kPa to get a steady flux, and then the pressure was changed to the operating pressure of 0.1 kPa and the water flux (Jw1) was calculated by the following equation:
where V was the volume of permeated water (L), A was the effective membrane area (m2) and ∆t was permeation time (h).
After switching the feed solution to 1.0 g/L BSA solution, BSA filtration was conducted for 1 h and the flux (Jp) was measured at 0.1 MPa. Finally, the membrane was cleaned with deionized water for 30 min and the pure water flux (Jw2) was measured again. The value Jw2/Jw1 represents the relative pure water performance after protein fouling and is used as a measurement of protein fouling for the dynamic flow studies.
Static protein adsorption experiments
The PVDF/GO-lysine membranes were cut into rectangular shape (2 cm × 1.6 cm) and then washed with PBS solution for 12 h and then were put into tubes filled with 10 mL BSA solutions in PBS solution with concentrations of 0.4 mg/mL and 0.6 mg/mL, respectively. The pH value of the PBS was adjusted to 7.4 in the experiments. At such a pH value, BSA was negatively charged (ζ ~ -22.3 mV). These tubes were incubated into a (25 ± 1) ℃ water bath for 24 h to reach equilibrium. The amount of protein adsorbed on the membrane was calculated by comparing the absorption intensity variation at 280 nm recorded by a UV-Vis spectrometer (HP 8453, USA).
RESULTS AND DISCUSSION
Preparation of the GO-amino Acids
At first, GO was prepared by introducing carboxylic acid, hydroxy and epoxy functional groups between carbon layers of graphite through strong oxidation process (Fig. 1). Based on these functional groups, the resulting GO can be further functionalized with amino acids which are cheap and environmental friendly. As shown in Fig. 1, the condensation reaction occurred between the amine groups of amino acids and carboxylic acid groups on the GO surface.
Figure 2 shows the dispersibility digital photographs of GO and GO-amino acids in water and organic solution, respectively. The dispersion of GO and GO-amino acids was both homogeneous at beginning in both water and organic solvent. After one day, the GO had a tendency to aggregate and was deposited at the bottom. Compared to GO, there was no obvious subsidence of GO-amino acids, especially of the GO-lysine. One month later, homogeneous and stable dispersion was observed for GO-lysine in both water and organic solvent. And GO and other GO-amino acids showed subsidence to some extent. In a word, GO-lysine kept a good dispersion stability even after a long time in both water and NMP, which made it a better choice as an additive to modificate the virgin PVDF membrane.
Characterization of the GO-amino Acids
Structure and Performance Characterization of PVDF/GO-lysine Membranes
XPS analysis
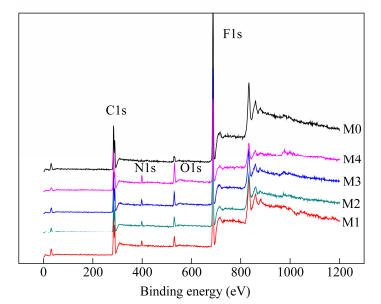
The element of the hybrid membranes was analysed by XPS in detail. Figure 8 showed the wide survey XPS spectra of virgin PVDF membrane (M0) and PVDF/GO-lysine membranes. Only C and F elements and trace amount of O element were observed in the survey spectrum of virgin PVDF membrane, corresponding to C1s (binding energy, 284.39 eV) and F1s (binding energy, 686.31 eV) for PVDF, O1s (530.27 eV) for PVP, respectively. In contrast with virgin PVDF membrane, there was a new peak of N1s at 398.25 eV for the PVDF/GO-lysine membranes. And the peak intensity of O1s and N1s was enhanced with the increase of the GO-lysine mass fraction from M1 to M4 gradually. This confirmed that more hydrophilic amino and carboxyl groups migrated to the membrane surface with the increase of GO-lysine mass fraction during the process of membrane preparation. And the migration made the peak intensity of N1s and O1s on membranes surface increased, which was conducive to improve the membrane surface performance.
Thermal stability
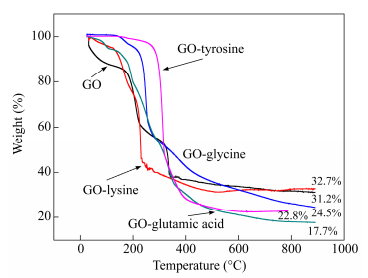
In this study, TGA analysis was used to determine the thermal stability of GO and GO-amino acids, and the results are shown in Fig. 7. For GO curve, a slight weight loss around 100 ℃ was attributed to the removal of adsorbed H2O molecules by O-containing functional groups tightly bounded into GO stacked structure. In addition, the weight loss around 250 ℃ was attributed to the decomposition of epoxy groups (C―O―C) on the GO surface, yielding CO, CO2, and steam[37-40]. And the loss around 400 ℃ to 500 ℃ may be attributed to the loss of carboxyl (C=O) and carboxylic groups (O―C=O)[41, 42]. For GO-amino acids, the total weight loss was 67.3% (GO-lysine), 75.5% (GO-glycine), 82.3% (GO-glutamic acid) and 77.2% (GO-tyrosine) respectively, which was related to the pyrolysis of organic moieties on the graphene oxide sheets. Although the grafting rate of lysine modified GO was relatively low, the thermal stability was better than others obviously.
Morphology of the PVDF/GO-lysine membranes
The surface SEM images of the pristine PVDF membrane and PVDF/GO-lysine membranes are presented in Fig. 9. Pores with 20-50 nm in diameter were observed on the surface for M0 (A0). After the GO-lysine was added into the PVDF membrane, pore size and porosity were increased slightly for M1, M2, M3 and M4 (B0, C0, D0 and E0, respectively).
For the increased pore size and porosity, the presence of the amphiphilic component in the casting solution may be expected to increase the affinity of casting solution and precipitant, enhancing solvent-nonsolvent exchange and creating the conditions for instantaneous de-mixing and associated macrovoid formation. Large macrovoids in the substructure typically result in the increased permeability, which are unsuitable for highpressure applications[43].
For the cross section of the membranes, all membranes presented asymmetric and highly inhomogeneous structure with a selective thin microporous upper layer on large voids. This structure was mainly due to the high mutual diffusivity of water and NMP[39]. Furthermore, the PVDF/GO-lysine membranes exhibited a better finger-like structure. This was because that the affinity of GO-lysine increases the mass transfer rate between solvent and non-solvent during phase-inversion process. The structure character of PVDF/GO-lysine membranes undoubtedly benefited the improvement of their water permeability.
XRD characterization
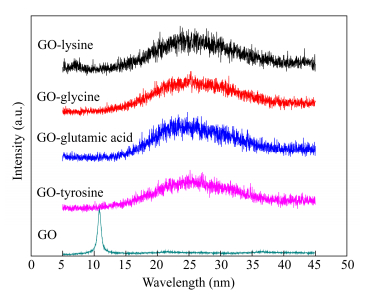
Figure 4 shows the XRD patterns of GO and variety of GO-amino acids. Obviously, the XRD pattern of GO showed a strong and sharp peak at 10.8°, which was different with the noticeable peak of graphite at 26.6°[34]. For the XRD patterns of GO-amino acids, the diffraction peak was obviously shifted to a higher diffraction angle compared to GO, indicating a decrease in the interplanar spacing determined from Bragg's law (2dsinθ = nλ). Specifically, the diffraction peak for GO-amino acids (Lysine, glycine, glutamic acid and tyrosine) shifted back to the original peak at 25°. The XRD pattern revealed that GO-amino acids demonstrated a particular crystal structure between that of graphite and graphene oxide.
While the mechanism for this unpredicted phenomenon maybe has not been reported. We inferred that the shift in the interlayer spacing is attributed to the reducing ability of amino acids. Present studies showed that amino acid was widely used as a reducing agent to prepare metal nanoparticles[35]. In this study, the condensation reaction between GO and amino acids resulted in the decrease of functional groups on GO surface. And the graphene oxide was reduced by amino acids in a certain extent.
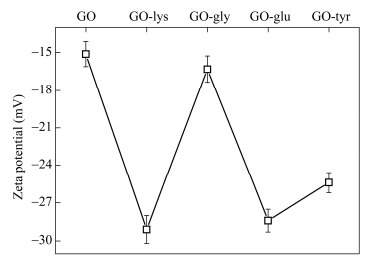
Zeta potential test
Zeta potential has been used for the characterization of the dispersion of GO-amino acids suspension. It should be noted that zeta potential can be realated to the stability of colloid dispersion, and a high zeta potential will confer stability[36]. As can be seen from the Fig. 6, the zeta potential was about -15.1 mV for GO. And it was -29.1 mV, -16.3 mV, -28.4 mV and -25 mV for GO-lysine, GO-glycine, GO-glutamic acid and GO-tyrosine, respectively. In short, the absolute value of the zeta potential was increased after the GO functionalized with amino acids, which suggested that dispersion stability of the GO-amino acids was improved greatly.
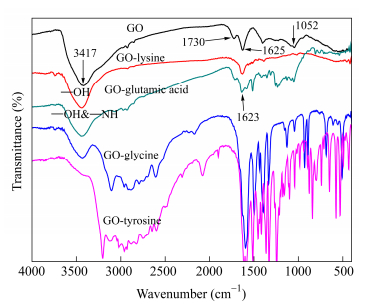
FTIR analysis
FTIR measurement was employed to investigate the chemical composition of the GO-amino acids. In the GO spectrum (Fig. 3), there were three characteristic peaks. The peak at 1052 cm-1 was attributed to the C―O bond. The peak at 3417 cm-1 was attributed to the O―H stretching bands of hydroxy and carboxylic acid moieties. And the presence C=O peak at 1730 cm-1 was attributed to C=O stretching band of carboxylic acids. These three peaks confirmed the presence of oxygen containing functional groups on GO surface after the oxidation process. For the spectra of different GO-amino acids, the C=O stretching of amide and carboxylate groups at the range of 1620-1630 cm-1 and the O―H and N―H stretching peaks at 3420 cm-1 confirmed the covalent functionalization of the GO by amino acids molecules successfully.
Hydrophilicity of the PVDF/GO-lysine membranes
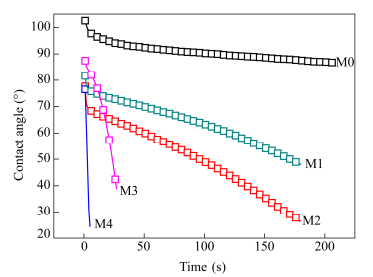
Hydrophilicity of the membrane surface is one of the most important factors affecting the protein fouling resistance of membranes. Static water contact angle measurements were employed to assess the wetting characteristics of the membrane surface (Fig. 11). The pristine PVDF membrane (M0) had the highest initial contact angle of 103°, corresponding to the lowest hydrophilicity. For GO-lysine amino modified membranes, the initial contact angles were remarkably decreased to 81.6° (M1), 77.7° (M2), 87.5° (M3) and 76.5° (M4), respectively. This means that the hydrophilicity of the PVDF/GO-lysine was improved significantly compared to that of the virgin PVDF membrane.
With the extension of time, descent velocity of the contact angle was obviously different for these membranes. In brief, the descent velocity of the contact angle for PVDF/GO-lysine was faster than that of the pristine PVDF membrane. For PVDF/GO-lysine membranes, increasing mass fraction of the GO-lysine made a quick decline of the contact angle descent curve. And the contact angle decreased to 10° within 6 s for M4. As well as for M3, it decreased to 37° within 26 s. All these demonstrated that the hydrophilicity of PVDF membrane was improved greatly with the addition of GO-lysine.
Antifouling property of PVDF/GO-lysine membranes
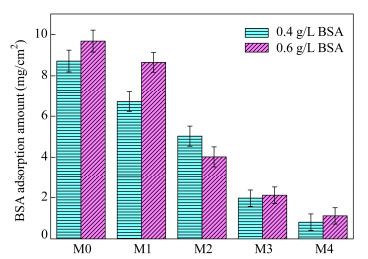
The protein resistance of the PVDF membrane and the modified membranes were tested under both static exposure and dynamic flow conditions. Figure 12 shows the BSA (0.4 mg/mL and 0.6 mg/mL) adsorbed amount onto the PVDF/GO-lysine membrane surface under static exposure conditions. Seen from the results, the nascent PVDF membrane had a high BSA adsorption of 9.69 mg/cm2 and 8.72 mg/cm2, respectively. Obviously, the PVDF/GO-lysine membranes showed small amount of BSA adsorption compared to the pristine PVDF membrane. The BSA adsorbed amount on the PVDF/GO-lysine surface was decreased with the increase of GO-lysine mass fraction in membrane casting solutions. And the PVDF/GO-lysine-8% showed a much less adsorbed BSA amount, which was only 10% of that of pristine PVDF membrane. In the case of the PVDF/GO-lysine membranes, a relatively hydrophilic membrane surface can keep the protein molecules away from contacting the hydrophobic PVDF directly. Therefore, most of the protein molecules depositing on the membrane surface can be easily removed off by water washing. These results were in consistence with the hydrophilic studies.
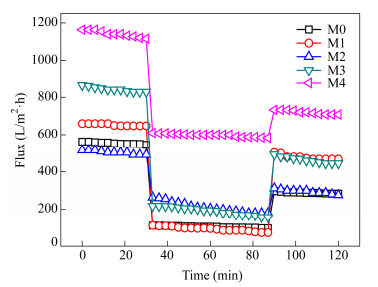
Ultrafiltration experiments were performed to further explore the protein fouling property of the PVDF/GO-lysine membranes under dynamic flow conditions. Figure 13 shows the permeation performance of these membranes. For Fig. 13, the water flux of the PVDF/GO-lysine membranes increased to a certain extent compared to the pristine PVDF membrane. After the filtrate was changed to BSA solution, the flux was lower than that of the pure water for all membranes. The distinct decrease in flux was caused by both fouling effects and concentration polarization effects[44]. The flux of the BSA solution for PVDF/GO-lysine-8% was higher than that of other membranes, corresponding to less protein fouling under static exposure conditions and contact angle. After protein filtration operation, the membranes were rinsed with deionized water and the water flux was measured again. Due to the irreversible adsorption of protein on the membrane surface and the entrapment of protein within membrane pores, the water flux could not recover to the original values. And the flux recovery ratios for M1, M2, M3 and M4 were 73.81%, 58.32%, 46.04% and 51.95%, respectively. It indicated that the PVDF/GO-lysine-2% membrane had superior resistance to protein fouling.
For mechanism of the fouling resistance, one was that the lysine is a basic amino acid which contains two amino groups and one carboxylic group. At neutral pH, amino and carboxylic group were protonated and deprotonated respectively. After consuming one amino group in the functional procedure, lysine grafted on GO could be regarded as zwitterionic groups. Another was that the electronic interaction plays a key role on the membrane interaction with proteins. The zeta potentials of the PVDF/GO-lysine membranes were higher than that of the virgin PVDF membrane. This means that the electrostatic repulsion of the PVDF/GO-lysine membranes and protein is higher than that of the virgin PVDF membrane and protein, which improved the antifouling properties of the PVDF/GO-lysine membranes. Therefore, it should be not surprising that lysine in membranes could confer high protein resistance to membrane surface. Besides, It should be noted that GO-lysine was charged material, which may not be good antifouling material for protein at low pH according to the “Whitesides rules” since their antifouling properties can be highly pH dependent.
Zeta potential of the PVDF/GO-lysine membranes
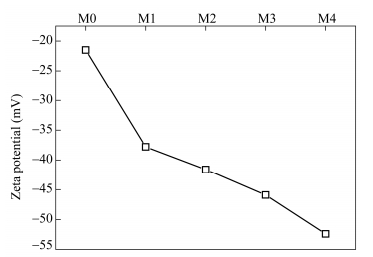
The addition of the GO-lysine to the PVDF membrane can lead to the alteration of the surface electrical property of the modified membrane. Figure 10 shows the experimentally measured zeta potentials of the membrane surfaces with different mass fractions of GO-lysine. The zeta potential of the virgin PVDF membrane was about -20.5 mV. With the additon of GO-lysine, the the zeta potentials of the PVDF/GO-lysine membrane surfaces showed a stronger negative zeta potential. Especially, the zeta potential reached to about -52.6 mV for PVDF/GO-lysine-8% membrane. The different electrical properties of the PVDF/GO-lysine membranes can have an effect on the anti-biofouling performance of the prepared membranes.
Morphology studies
TEM measurement was performed to observe the morphological structure of GO and GO-amino acids, which is shown in Fig. 5. We can distinctly find that GO exhibits a natural tendency to unfold and presents a flake-like and wrinkled morphology resembling the exfoliated crumpled thin flake. And the GO-amino acids possessed clear crystal structure compared to GO, which was corresponding with the XRD results. The crystal structure was more obvious for GO-lysine and GO-tyrosine and the lattice fringe was clear under high magnification TEM. Based on the TEM results above, we can conclude that the GO-amino acids exhabited crystal structure obviously compared to GO.
CONCLUSIONS
In this study, functionalized GO was prepared by the covalent functionalization with amino acids (lysine, glycine, glutamic acid and tyrosine). FTIR and XPS results demonstrated that amino acids were successfully functionalized onto GO surface. At the same time, its disperision stability in water and organic solvents was improved greatly. GO-lysine was chosen to prepare the PVDF/GO-lysine membranes with different mass fractions. The hydrophilicity was improved significantly with the contact angle of 75° compared to that of nascent PVDF membrane. The zeta potential absolute value of the PVDF/GO-lysine membranes was higher than that of the virgin PVDF membrane. And the PVDF/GO-lysine membranes displayed high resistance to protein adsorption and protein fouling from aqueous solution under both static exposure and dynamic filtration conditions. In view of the above results, the superior performance of the functionlalized hybrid membranes offers a great poretial for pratical application.
-
-
[1]
Li, J.H., Shao, X.S., Zhou, Q., Li, M.Z. and Zhang, Q.Q., Appl. Surf. Sci., 2013, 265: 663
-
[2]
Mahdie, S.K. and Vahid, V., Ind. Eng. Chem. Res., 2014, 53: 13370
-
[3]
Liu, J., Shen, X., Zhao, Y.P. and Chen, L., Ind. Eng. Chem. Res., 2013, 52: 18392
-
[4]
Chia-Hung, K., Chen, G.J., Yawo-Kuo, T., Liu, Y.C. and Chwen-Jen S., Ind. Eng. Chem. Res., 2012, 51: 5141
-
[5]
Wang, J.H., Zhang, Y.H., Xu, Y.Y. and Zhu, B.K., Chinese J. Polym. Sci., 2014, 32(2): 143
-
[6]
Lee, N., Amy, G., Croué, J.P. and Buisson, H., Water Res., 2004, 38: 4511
-
[7]
Xu, Z.W., Zhang, J.G., Shan, M.J., Li, Y.L., Li, B.D. and Niu, J.R., J. Membr. Sci., 2014, 458: 1
-
[8]
Nabe, A., Staude, E. and Belfort, G., J. Membr. Sci., 1997, 133: 57
-
[9]
Schulze, A., Maitz, M.F., Zimmermann, R., Marquardt, B., Fischer, M., Werner, C., Wenta, M. and Thomas, I., RSC Adv., 2013, 3: 22518
-
[10]
Meng, J.Q., Chen, C.L., Huang, L.P., Du, Q.Y. and Zhang, Y.F., Appl. Surf. Sci., 2011, 257: 6282
-
[11]
Yuan, T., Meng, J.Q., Hao, T.Y., Wang, Z.H. and Zhang, Y.F., ACS Appl. Mater. Interfaces, 2015, 7: 14896
-
[12]
Yuan, J., Meng, J.Q., Kang, Y.L., Du, Q.Y. and Zhang, Y.F., Appl. Surf. Sci., 2012, 258: 2856
-
[13]
Sun, Q., Zhang, Y.F., Chen, C.L., Guo, X.Z. and Meng, J.Q., Chinese J. Polym. Sci., 2014, 32(7): 880
-
[14]
Singh, A.K., Singh, P., Mishra, S. and Shahi, V.K., J. Mater. Chem., 2012, 22: 1834
-
[15]
Majeed, S., Fierro, D., Buhr, K., Wind, J., Du, B., Boschetti-de-Fierro, A. and Abetz, V., J. Membr. Sci., 2012, 403-404: 101
-
[16]
Sui, Y., Wang, Z.N., Gao, X.L. and Gao, C.J., J. Membr. Sci., 2012, 413-414: 38
-
[17]
Venault., A., Liu, Y.H., Wu, J.R., Yang, H.S., Chang, Y., Lai, J.Y. and Aimar, P., J. Membr. Sci., 2014, 450: 340
-
[18]
Wu, M.Y., Meng, S.J., Wang, Q., Huang, W. and Dong, X.C., ACS Appl Mater Inter., 2015, 7: 21089
-
[19]
Xia, S.J. and Ni, M.Z., J. Membr. Sci., 2015, 473: 54
-
[20]
Cao, K.T., Jiang, Z.Y., Zhao, J., Zhao, C.H., Gao, C.Y., Pan, F.S., Wang, B.Y., Cao, X.Z. and Yang, J., J. Membr. Sci., 2014, 469: 272
-
[21]
Zhu, Y.W., Murali, S., Cai, W.W., Li, X.S., Suk, J.W., Jeffrey, R.P. and Rodney, S. R., Adv. Mater., 2010, 22: 3906
-
[22]
Konkena, B. and Vasudevan, S., Langmuir., 2012, 28: 12432
-
[23]
Konkena, B. and Vasudevan, S., J. Phys. Chem. C., 2015, 119: 6356
-
[24]
Loh, C.H. and Wang, R., J. Membr. Sci., 2013, 446: 492
-
[25]
Birkner, M. and Ulbricht, M., J. Membr. Sci., 2015, 494: 57
-
[26]
Yang, R., Goktekin, E. and Karen K. G., Langmuir., 2015, 31: 11895
-
[27]
Duan, X.B. and Randy S. L., Biomaterial., 2002, 23: 1197
-
[28]
Shi, Q., Su, Y.L., Chen, W.J., Peng, J.M., Nie, L.Y., Zhang, L. and Jiang, Z.Y., J. Membr. Sci., 2011, 366: 398
-
[29]
Wang, J., Yao, Y., Ji, B., Huang, W., Zhou, Y.F. and Yan, D.Y., Chinese J. Polym. Sci., 2011, 29(2): 241
-
[30]
Jayalakshmi, A., Rajesh, S. and Mohan, D., Appl. Surf. Sci., 2012, 258: 9770
-
[31]
Lapointe, J.F., Gauthier, S.E., Pouliot, Y. and Bouchard, C., J. Membr. Sci., 2005, 261: 36
-
[32]
Dinh K.D. and Euij, K., Nanoscale Re Lett., 2015, 10: 6
-
[33]
Daniela, C.M., Dmitry, V.K., Jacob, M.B., Alexander, S., Sun, Z.Z., Alexander, S., Lawrence, B.A., Lu, W. and James, M.T., ACS Nano., 2010, 8 : 4806
-
[34]
Liu, H.Y., Cheng, J., Chen, F.J., Hou, F.P., Bai, D.C., Xi, P.X. and Zeng, Z.Z., ACS Appl. Mater. Interfaces., 2014, 6: 3132
-
[35]
Kuilla, T., Bhadra, S., Yao, D., Kim, N.H., Bose, S. and Lee, J.H., Prog. Polym. Sci., 2010, 35: 1350
-
[36]
Callejas Fernández, J., de las Nieves, F.J., Martínez García, R. and Hidalgo-Alvarez, R., Colloids Surf., 1991, 61: 123
-
[37]
Kong, J.Y., Choi, M.C., Kim, G.Y., Park, J.J., Selvaraj, M., Han, M. and Ha, C.S., Eur. Polym. J., 2012, 48: 1394
-
[38]
Compton, O.C., Dikin, D.A., Putz, K.W., Brinson, L.C. and Nguyen, S.T., Adv. Mater., 2010, 22: 892
-
[39]
Yang, Y. and Liu, T., Appl. Surf. Sci., 2011, 257: 8950
-
[40]
Luan, V.H., Tien, H.N. and Hur, S.H., J. Colloid Interface Sci., 2015, 6: 437
-
[41]
Luan, V.H., Tien, H.N., Hoa, L.T., Hien, N.T.M., Oh, E.S. and Chung, J., J. Mater. Chemistry A., 2013,1: 208
-
[42]
Hester, J.F., Banerjee, P., Won, Y.Y., Akthakul, A., Acar, M.H. and Mayes. A.M., Macromolecules., 2002, 35: 7652
-
[43]
Wang, Z., Yu, H., Xia, J., Zhang, F., Li, F., Xia, Y. and Li, Y., Desalination., 2012, 299: 50
-
[44]
Shi, Q., Su, Y.L., Chen, W.J., Peng, J.M., Nie, L.Y., Zhang, L. and Jiang, Z.Y., J. Membr. Sci., 2011, 366: 398
-
[1]
-

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 0
- 文章访问数: 1756
- HTML全文浏览量: 40




 下载:
下载:

