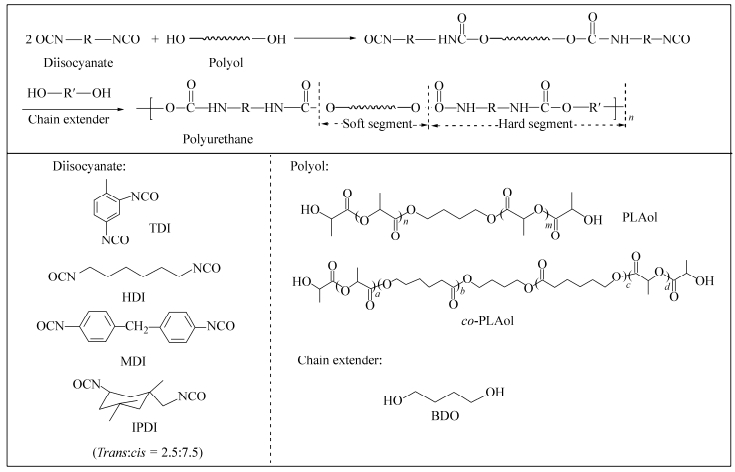
 Figure Scheme1.
Synthetic route of the bio-PUs
Figure Scheme1.
Synthetic route of the bio-PUs

High Performance Bio-based Polyurethane Elastomers: Effect of Different Soft and Hard Segments
English
High Performance Bio-based Polyurethane Elastomers: Effect of Different Soft and Hard Segments
-
Key words:
- Polylactide diols
- / Bio-based polyurethane
- / Shape memory
- / Biocompatibility
-
INTRODUCTION
Currently, a great number of disposable medical devices, such as infusion apparatus, catheters, and blood bag are used to avoid the inflection from medical devices[1]. Most of these single-use medical devices are made from phthalate plasticized poly(vinyl chloride) (PVC), but the plasticizers often cause troubles, especially upon contact with body fluids or tissues[2, 3]. Some polyolefin alloys or thermoplastic elastomers have been reported to partially replace soft PVC in medical devices[1, 2, 4]. However, the above-mentioned materials are not biodegradable, which would result in environmental pollution over a long period of time. Therefore, it is desired to develop a green material as an alternative to PVC in medical devices.
On the other hand, significant interest has been paid to biodegradable polymers due to serious environment pollution and their biomedical applications[5-8]. Among them, polylactide (PLA), produced from bio-based lactic acid, is known to be one of the promising polymers because of its good biodegradability and biocompatibility[9-11]. It can be degraded into small molecules in the body, and then further broken down into CO2 and water, which can be excreted through metabolism. However, the brittle nature, poor ductility and impact toughness of PLA limit some applications where mechanical toughness is required[12, 13]. Recently, a series of PLA-based polyurethanes (PUs) with shape memory properties have been reported[14-17]. For example, Jing et al. reported PLA-based PUs with glass transition temperature (Tg) in the range of 33-63℃were synthesized from PLA diols, diisocyanate compounds and 1, 4-butanediol (BDO)[15, 16]. However, these PLA-based PUs showed high Young’s modulus and hardness, which makes them unsuitable to replace PVC in medical devices.
In the present work, a series of high performance bio-based polyurethanes (bio-PUs) were synthesized from PLA-based diols, different diisocyanates and chain extender 1, 4-butanediol, in which different soft and hard segments are used to adjust their transition temperatures and mechanical properties. Poly(lactide-co-caprolactone) copolymer diols instead of PLA diols as the soft segment improved the thermal stability and mechanical properties of the synthesized bio-PUs. Among them, MDI-based bio-PUs exhibit better mechanical properties than soft PVC, closed to petroleum-based commodities. Furthermore, the obtained bio-PUs display good shape memory properties at body temperature and cytocompatibility. Therefore, these bio-PUs are promising for applications in biomedical fields, which are especially expected to replace PVC in medical devices.
EXPERIMENTAL
Materials
Hexamethylene diisocyanate (HDI), 4, 4 -diphenyl methane diisocyanate (MDI), 1, 4-butanediol (BDO) and stannous octoate (SnOct2) are analytically pure, and were obtained from Aladdin (Shanghai, China). Toluene diisocyanate (TDI), tetrahydrofuran (THF) and N, N-dimethyl formamide (DMF) were purchased from Sinopharm Chemical Reagent Co., Ltd. e-Caprolactone (CL) and L-lactide (LA) were provided from Shenzhen Esun Industrial Co., Ltd.
Synthesis of Bio-based PLA and Copolymer Diols
A series of PLA diols (PLAols) were synthesized by ring opening polymerization of LA using SnOct2and BDO as the catalyst and chain transferring agent, respectively. In a N2 atmosphere, LA, BDO and SnOct2 were added to a 1-L flask free of oxygen and water, where the catalyst amount was 0.1% with respect to LA, wt/wt. The reaction was performed at 160℃for 6 h. The molecular weight of PLAols was tuned to be 1000 and 2000 g/mol by changing the feed molar ratio of BDO and LA (1/6.0, 1/13.3), which were denoted as PLAol-1000 and PLAol-2000, respectively.
The poly(LA-co-CL) diols (co-PLAols) were prepared in a similar way, where the feed molar ratio of LA and CL was 3.17. The co-PLAols with molecular weight of 1000, 2000 and 3000 g/mol were nominated as co-PLAol-1000, co-PLAol-2000 and co-PLAol-3000, respectively.
Preparation of PLA-based Polyurethanes
PLA-based polyurethanes were synthesized via a two-step polymerization process, where the molar ratio of polyol, diisocyanate and chain extender is 1:2:1, as shown in Scheme 1. First, the polyol was dissolved in THF at 60℃and then diisocyanate and SnOct2were added to the THF solution. Second, when the remaining NCO content was closed to the theoretical value, the chain extender was added to the above reaction system. The reaction was carried out until the FTIR absorption peak of NCO group (2270 cm-1) disappeared. The detailed information of the obtained bio-PUs are listed in Table 1.
Samples Polyols Diisocyanates HSa (wt%) Mwb Mw/Mnb T1K PLAol-1000 TDI 29.2 1.7 x 104 2.1 T2K PLAol-2000 TDI 18.1 2.0 x 104 2.1 co-T1K co-PLAol-1000 TDI 30.5 1.7 x 104 2.0 co-T2K co-PLAol-2000 TDI 18.0 2.2 x 104 2.0 co-T3K co-PLAol-3000 TDI 12.7 2.3 x 104 2.2 co-H2K co-PLAol-2000 HDI 17.5 2.2 x 104 2.0 co-M2K co-PLAol-2000 MDI 22.8 3.5 x 104 2.3 co-I2K co-PLAol-2000 IPDI 21.1 2.7 x 104 2.1 a The hard segment weight percent (HS, wt%), which is defined as the percent by weight of BDO and diisocyanates in the polyurethane; b Measured by GPC Table 1. Detail information of the obtained bio-PUsCharacterization Methods
FTIR spectra were performed on a Thermo Nicolet 6700 spectrometer under an attenuated total reflection (ATR) mode. 1H-NMR spectra were measured at room temperature on a Bruker AV-400 NMR apparatus in deuterated chloroform (CDCl3). The molecular weights (Mn, Mw) of the resulted polyurethanes were obtained on a HLC-8320 gel permeation chromatography (GPC) according to polystyrene (PS) standard using THF as eluent.
Differential scanning calorimetry (DSC) analysis was recorded using a DSC 214 of Polyma (Netisch) Instruments. The samples were initially heated from −30℃to 180℃, then cooled to −30℃, and finally heated up to 180℃at a rate of 10 K/min in a nitrogen atmosphere. The glass transition temperature (Tg) was determined from the second heating scan to reduce the influence of thermal history. Thermo gravimetric analysis (TGA) was performed using a TGA-Q50 system from TA Instruments at a heating rate of 20 K/min in a nitrogen atmosphere. Atomic force microscopy (AFM) was conducted on a Dimension 3100 V scanning probe microscope in tapping mode. The AFM sample was prepared by casting the bio-PUs solution (5 wt%) on a silicon wafer.
Tensile tests were conducted on a tensile instrument (Instron 5567) with a speed of 100 mm/min at 25℃. The mean value of five replicated measurements was taken for each sample. The shape memory test of bio-PUs was performed as follows: Firstly, the specimens were bent to a given angle at 37℃. Subsequently, they were quenched below Tg using liquid nitrogen. Then the samples were allowed for free recovery at 37℃.
Cytotoxicity of bio-PUs was examined by Alamar blue assay. After sterilization via sonication for 30 min in DI water and 75% ethanol respectively, the specimens were immersed in Eagle’s minimum essential medium, Alpha modification (a-MEM; Gibco) supplemented with 10% fetal bovine serum (FBS; Gibco), 1% penicillin and 1% streptomycin, and incubated for 72 h under the cell culture conditions (95% humidity, 5% CO2, 20% O2 37℃). 2×105 of human mesenchymal stem cells (hMSCs) were seeded in each well of a 24-well plate. The cell viability was examined by Alamar blue assay at several time points after incubation in the extracts (days 1 and 3) and compared with the viability in the pure cell culture medium. Briefly, 50μL of Alamar blue assay reagent were added into each well of the culture plate and the absorbance at 562 nm was measured after incubation for 4 h in dark. Each group consisted of 4 independent samples.
RESULTS AND DISCUSSION
Properties of Bio-PUs
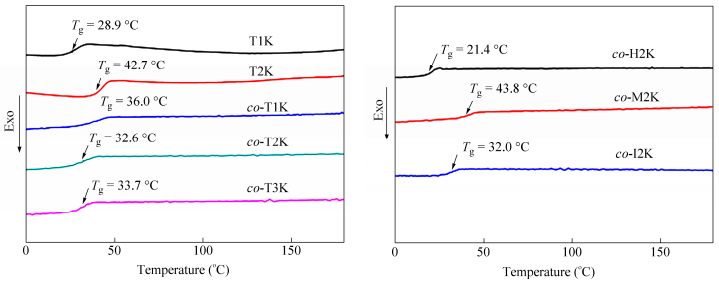
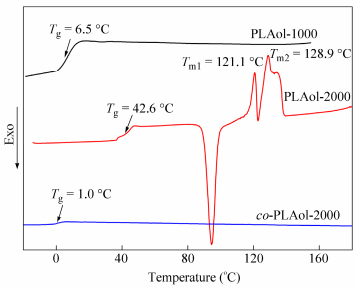
Figure 4 shows the DSC curves of the synthesized bio-PUs with different soft and hard segments, and their Tg values are summarized in Table 2. All obtained bio-PUs display only one Tg and no crystallization or melting peaks in the DSC curves, indicating that the obtained PUs were amorphous[19]. As the molecular weight of soft segments PLAols increased from 1000 to 2000, the Tg values of TDI-based PUs increased from 28.9℃to 42.7℃. While co-PLAols were used to be the soft segments, the Tg values of TDI-based PUs were very closed. The PU synthesized from MDI has a higher Tg than the TDI-based one, and the reason lies in that MDI has more symmetrical structure than TDI, which may help to enhance the intermolecular interaction, leading to higher Tg. Moreover, the Tg values of the synthesized bio-PUs are closed to body temperature. Hence, as shape memory polymers, these obtained bio-PUs are expected to have many applications in medical devices.
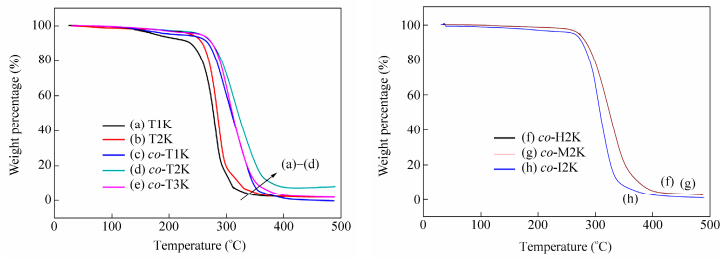
Tg (℃) T5% (℃) Tmaxa (℃) Tensile strength (MPa) Elongation (%) Young’s modulus (MPa) T1K 28.9 182.7 281.5 - - - T2K 42.7 239.7 288.0 - - - co-T1K 36.0 214.9 293.6 - - - co-T2K 32.6 256.3 319.0 8.8 460 217.3 co-T3K 33.7 255.4 307.3 5.6 440 84.1 co-H2K 21.4 270.8 329.4 2.2 580 22.0 co-M2Kco-I2K 43.8 32.0 274.1 261.9 302.2 306.4 23.5 3.5 430 350 380.8 137.5 a Tmax is the temperature of the maximum rate of weight-loss of the samples. Table 2. Thermal and mechanical properties of bio-PUs samplesThe effect of different soft and hard segments on the thermal stability of the obtained bio-PUs was investigated by TGA, as shown in Fig. 5. Their weight loss temperatures (T5%, Tmax) are listed in Table 2. Instead of PLAols as the soft segment, co-PLAols improved the thermal stability of the synthesized PUs. In comparison, the type of diisocyanates has a little impact on the thermal stability of the obtained PUs. Furthermore, the T5% and Tmax of the bio-PUs synthesized from co-PLAols are above 200 and 300℃, respectively, indicating the good thermal stability of these bio-PUs.
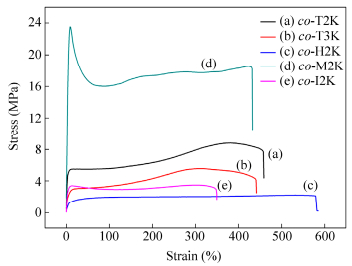
Figure 6 displays the typical stress-strain curves of the obtained bio-PUs from co-PLAols, and their tensile strength, elongation and Young’s modulus are summarized in Table 2. Obvious yielding was observed before break. From Table 2, the tensile strength and modulus of the samples decreased with the molecular weight of soft segment increasing, from 8.8 MPa and 217.3 MPa for co-T2K to 5.6 MPa and 84.1 MPa of co-T3K, respectively. Compared with TDI, MDI and IPDI-based PUs, HDI-based PUs have the highest elongation at break, possibly due to the long aliphatic carbon chains of HDI, resulting in good flexibility of the PU chains. From Fig. 6 and Table 2, we can conclude that the mechanical properties of the obtained bio-PUs could be adjusted in a wide range. The tensile strength and elongation of co-M2K synthesized from co-PLAol-2000 and MDI were 23.5 MPa and 430%, respectively, which are comparable to commercial petroleum-based polyester PUs (Desmopan® 400 serials) from Bayer Corporation[20]. The superior mechanical properties are mainly attributed to the well-defined phase separation morphology, as shown in Fig. 7. Compared with TDI, HDI and IPDI-based PUs, the micro-phase of MDI-based PUs becomes more developed.
The shape memory properties of the obtained bio-PUs at body temperature are also measured. Figure 8 shows the shape recovery process of co-I2K at body temperature (37℃). The sample was changed into a bending shape at 37℃and then cooled rapidly below Tg using liquid nitrogen and it reached the original shape state in about 2 min at 37℃.
The above-mentioned results indicate that the synthesized bio-PUs display better mechanical properties and good shape memory properties at body temperature, which are promising for applications in medical devices.
Cytotoxicity of Bio-PUs
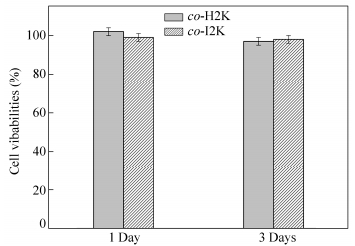
To determine the cytotoxicity of the synthesized bio-PUs, the Alamar blue assay by hMSCs was used, as shown in Fig. 9. It can be clearly seen that co-H2K and co-I2K showed no cytotoxicity, and their cell vibabilities were nearly closed to 100% after incubation for 1 and 3 days. This result indicates that the synthesized bio-PUs are cytocompatible to hMSCs[21].
Synthesis and Characterization of Bio-based PLA Diols and Polyurethanes
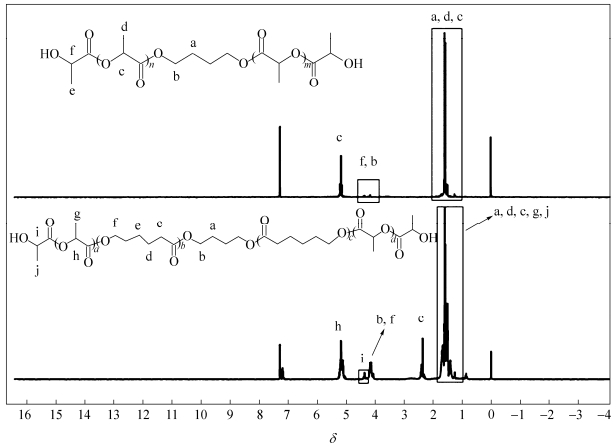
Five bio-based PLA and copolymer diols (PLAol-1000, 2000 and co-PLAol-1000, 2000, 3000) were synthesized by the above-mentioned method. The structures of PLAol and co-PLAol were confirmed by 1H-NMR, as shown in Fig. 1. The signals at d=5.17 are ascribed to the―CH―connected to C=O in PLAol and co-PLAol, while the signals at d=2.32, 1.64, 1.29, 1.62 and 4.08 are assigned to protons on―C=O―CH2―CH2―CH2―CH2―CH2―O―in co-PLAol, respectively[18]. Figure 2 shows the DSC curves of the synthesized PLAol and co-PLAol. PLAols with Mn of above 2000 have multi-melting peaks, and their Tg values increase with the increase of Mn. In comparison, co-PLAols are amorphous and have relatively low Tgs. Instead of an elastomer, the bio-PUs synthesized from PLAols and TDI were rigid solid at room temperature. To obtain high performance bio-PUs, co-PLAols were therefore chosen to be used as the soft segments.
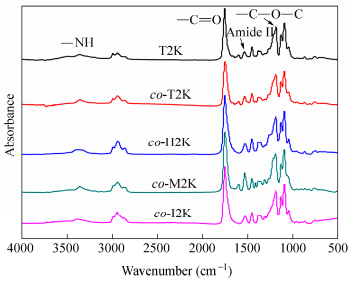
As shown in Scheme 1, the bio-PUs were prepared from different diisocyantes via a two-step polymerization process, whose structure was characterized by FTIR spectra. The spectra (Fig. 3) exhibit absorption peaks at about 3360 and 1530 cm-1, which are assigned to the stretching vibrations of NH and amide II in the urethane groups, and they do not display the stretching vibration bands of N=C=O groups at about 2270 cm-1 and OH groups at about 3500 cm-1. Moreover, the strong absorptions at about 1750 and 1200 cm-1 correspond to the stretching vibrations of C=O and C―O―C in the ester groups of polyols, respectively. These results demonstrate that the bio-PUs from different diisocyanates were successfully synthesized. In the following sections, the thermal, mechanical, shape memory properties and phase separation behavior of the synthesized bio-PUs with different hard segments would be discussed.
CONCLUSIONS
A series of high performance bio-PUs elastomers from PLA-based diols and different diisocyanates were successfully prepared and characterized. Instead of PLAols as the soft segment, co-PLAols improved the thermal stability and mechanical properties of the synthesized PUs. Among them, co-M2K from MDI demonstrates better mechanical properties, closed to the petroleum-based polyester polyurethane (Bayer Desmopan® 400 series). Moreover, the obtained bio-PUs display good shape memory properties at body temperature and biocompatibility, which are hence promising for applications in biomedical fields and especially expected to replace PVC for medical devices.
-
-
[1]
Ishihara, K., Nishiuchi, D., Watanabe, J. and Iwasaki, Y., Biomaterials, 2004, 25(6): 1115
-
[2]
Chiellini, F., Ferri, M., Morelli, A., Dipaola, L. and Latini, G., Prog. Polym. Sci., 2013, 38(7): 1067
-
[3]
Tickner, J.A., Schettler, T., Guidotti, T., McCally, M. and Rossi, M., Am. J. Ind. Med., 2001, 39(1): 100
-
[4]
Wang, W.S., Ping, P., Yu, H.J., Chen, X.S. and Jing, X.B., J. Polym. Sci., Part A: Polym. Chem., 2006, 44(19): 5505
-
[5]
Wang, Z.G., Yu, L.Q., Ding, M.M., Tan, H., Li, J.H. and Fu, Q.A., Polym. Chem., 2011, 2(3): 601
-
[6]
Gu, L., Gao, Y.G., Qin, Y.S., Chen, X.S., Wang, X.H. and Wang, F.S., J. Polym. Sci., Part A: Polym. Chem., 2013, 51(2): 282
-
[7]
Song, Z.M., Shi, B., Ding, J.X., Zhuang, X.L., Zhang, X.N., Fu, C.F. and Chen, X.S., Chinese J. Polym. Sci., 2015, 33(4): 587
-
[8]
Gu, L., Wu, Q.Y. and Yu, H.B., Chinese J. Polym. Sci., 2015, 33(6): 838
-
[9]
Shao, L.N., Dai, J., Zhang, Z.X., Yang, J.H., Zhang, N., Huang, T. and Wang, Y., RSC Adv., 2015, 5(123): 101455
-
[10]
Gu, S.Y. and Gao, X.F., RSC Adv., 2015, 5(109): 90209
-
[11]
Yan, B., Gu, S. and Zhang, Y., Eur. Polym. J., 2013, 49(2): 366
-
[12]
Zhang, L., Xiong, Z., Shams, S.S., Yu, R., Huang, J., Zhang, R. and Zhu, J., Polymer, 2015, 64: 69
-
[13]
Xing, Q., Li, R.B., Dong, X., Zhang, X.Q., Zhang, L.Y. and Wang, D.J., Chinese J. Polym. Sci., 2015, 33(9): 1294
-
[14]
Peponi, L., Navarro-Baena, I., Sonseca, A., Gimenez, E., Marcos-Fernandez, A. and Kenny, J.M., Eur. Polym. J., 2013, 49(4): 893
-
[15]
Wang, W.S., Ping, P., Chen, X.S. and Jing, X.B., Eur. Polym. J., 2006, 42(6): 1240
-
[16]
Wang, W.S., Ping, P., Chen, X.S. and Jing, X.B., Polym. Int., 2007, 56(7): 840
-
[17]
Xie, M., Wang, L., Ge, J., Guo, B. and Ma, P.X., ACS Appl. Mater. Inter., 2015, 7(12): 6772
-
[18]
Gu, L., Cui, B., Wu, Q.Y. and Yu, H., RSC Adv., 2016, 6(22): 17888
-
[19]
Zhang, C., Madbouly, S.A. and Kessler, M.R., ACS Appl. Mater. Inter., 2015, 7(2): 1226
-
[20]
Zhang, L., Huang, M., Yu, R., Huang, J., Dong, X., Zhang, R. and Zhu, J., J. Mater. Chem. A, 2014, 2(29): 11490
-
[21]
Liu, Y., Hu, J., Zhuang, X., Zhang, P., Wei, Y., Wang, X. and Chen, X., Macromol. Biosci., 2012, 12(2): 241
-
[1]
-

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 0
- 文章访问数: 2222
- HTML全文浏览量: 367



 下载:
下载:

