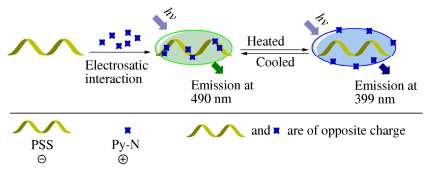
 Figure Scheme 1.
The mechanism of polymer fluorescent thermometer
Figure Scheme 1.
The mechanism of polymer fluorescent thermometer

Temperature-responsive Behavior of Polymer Fluorescent System via Electrostatic Interaction Mediated Aggregation/Deaggregation
English
Temperature-responsive Behavior of Polymer Fluorescent System via Electrostatic Interaction Mediated Aggregation/Deaggregation
-
INTRODUCTION
Environmental stimuli-responsive polymer systems have received great interest in recent years[1]. Among the environmental factors, temperature plays a significant role in different biological activities, as well as in chemistry and other fields[2-5]. For some precision instruments or therapeutic systems, temperature and its distribution are required to be kept monitoring. Various conventional thermometers can provide quantitative temperature measurements under general conditions; however, few of them can accurately measure temperature with high spatial resolution under complicated circumstance. For instance, it is still challenging for various conventional methods to measure temperature distribution of a system in the dark[4, 6]. Recently, fluorescence thermometers have been developed rapidly, which essentially inherit the merits of fluorescence sensing[7-14], showing great sensitivity toward temperature changes and flexibility in applications[15-18]. Some of them were designed by combining fluorescent chromophores with thermosensitive polymers such as poly(N-isopropylacrylamide) (PNIPAm), as the PNIPAm possesses lower critical solution temperature (LCST) in aqueous solution[19-26]. Others were molecular thermometers, taking advantage of temperature-dependent fluorescence signal variation[3, 27-30]. These fluorescent thermometers adopted non-contact optical approaches, thus they can be applied in measuring temperature under relatively severe environmental conditions. Compared with conventional thermometers, fluorescent thermometers have already exhibited great potential for practical applications under complicated circumstance[15, 31, 32].
Previous development of fluorescent thermometers is mainly based on fluorescent signal change induced by thermally stimulated environmental changes. Among these fluorescence sensors, a few utilized the excimer/monomer formation of pyrene derivatives, which usually incorporated two pyrene moieties into one molecule and functioned through temperature-induced changes of the distance between the two pyrene moieties[4, 6, 27]. Up to now, there have been no literature reports concerning fluorescent temperature sensors that is based on the combination of the excimer/monomer formation of pyrene derivatives and the electrostatic interactions in a polymer system. Pyrene, as a commonly-used fluorophore, with its monomer emission at 370-400 nm, is able to form intramolecular excimers (emission at 450-500 nm)[33, 34] when aggregated, and its excimer also can transform back to the monomer state after deaggregation, therefore various fluorescent probes have been designed based on this feature[35, 36].
We envision that, taking full advantage of polyelectrolyte’s electrostatic interaction and multiple anchoring sites along macromolecular chains could greatly facilitate the excimer/monomer conversion of charged pyrene derivatives; and the polymer system with the synergistic effect of excimer/monomer conversion and electrostatic interaction could eventually turn out to be a promising thermoresponsive system. As a proof of concept, herein, we developed a facile and effective fluorescent thermometer functioning via excimer/monomer interconversion of pyrene derivative and electrostatic interaction. The schematic illustration for the system is shown in Scheme 1. Poly(sodium 4-styrenesulfonate) (PSS)[37, 38], with high negative charge densities in its polymeric chains, was selected as a model polyelectrolyte, which served as the anchoring carrier for positively-charged pyrene derivative molecules (Py-N). In the system, the positively charged Py-N molecules were adsorbed along the negatively-charged polymer chain and readily formed aggregation in solution, displaying excimer emission. When the solution was heated, the Py-N molecules might get sufficient energy to break out of electrostatic attractions, thereby resulting in the disaggregation and restoring the monomer emission of pyrene derivative. The system herein is a novel and simple ratiometric polymer fluorescent system for temperature response, and shows the capacity of overcoming the interferences of single-intensity detection mode resulting from external factors like excitation intensity and sample concentration[39-43]. Furthermore, the system is technically-simple to prepare, can exhibit high spatial resolution, and is easy for long-range control.
EXPERIMENTAL
Materials and Characterization
1-Pyrenemethanamine hydrochloride, acetic anhydride N, N-diisopropylethylamine, bromoacetic acid, 1-[bis(dimethylamino)methylene]-1H-1, 2, 3-triazolo[4, 5-b]pyridinium 3-oxid hexafluorophosphate (HATU) and 30% trimethylamine solution were purchased from Aladdin Reagents and used as received. Poly(sodium 4-styrenesulfonate) (average Mw ~70000) was purchased from Sigma-Aldrich and used as received. MgSO4, NaH2PO4·2H2O and Na2HPO4·7H2O of analytical grade were purchased from Guangzhou Chemical Reagent Factory and used as received.
All solvents (acetonitrile, dichloromethane, acetone, methanol, dimethylsulfoxide, diethyl ether) were of analytical grade. The water used in the experiments was the triple-distilled water treated by ionexchange columns and then by a Milli-Q water purification system.
1H-NMR spectra were obtained on a BrukerAvance 600 MHz NMR spectrometer (acquisition software: Topspin; processing programs: MestReNova). Mass spectra were measured through a Bruker Esquire HCT Plusmass spectrometer. UV-Vis spectra were recorded by using a HitachiU-3010 UV-Vis spectrophotometer. Fluorescence spectra were measured by using a Hitachi F-4600 fluorescence spectrophotometer (excitation wavelength: 350 nm) with a LAUDA Alpha RA 8 thermostat. The quantum yields of this polymer fluorescent system were acquired according to reported literatures[44, 45]. The CIE1931 chromaticity coordinates were acquired from initial experimental data according to reported literature[46].
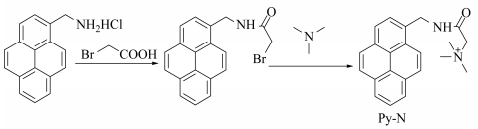
The synthetic routes of Py-N is shown in Scheme 2.
Synthesis of Py-N
Bromoacetic acid (83.4 mg, 0.6 mmol) was dissolved in 12 mL dichloromethane in 50 mL flask at room temperature. HATU (380.3 mg 1.2 mmol) was added to the solution and kept stirring for 30 min. 1-Pyrenemethanamine hydrochloride (133.9 mg, 0.5 mmol) and N, N-diisopropylethylamine (258.5 mg, 2 mmol) were dissolved in 4 mL dichloromethane. Then the solution was added into the flask. The reaction mixture was stirred in the dark overnight. Afterwards, the mixture was extracted by pure water and dichloromethane three times and the oil phase was dried by anhydrous MgSO4. The crude product was purified on a silica gel column chromatography (dichloromethane: methanol = 20:1) to yield a yellow powder (80 mg, yield 45%).
1H-NMR (600 MHz, DMSO-d6, δ): 9.02 (s, 1H), 8.35 (s, 1H), 8.28 (s, 4H), 8.18 (s, 2H), 8.09 (s, 1H), 8.07 (s, 1H), 5.05 (s, 2H), 3.36 (s, 2H).
The yellow powder (69.1 mg, 0.196 mmol) was dissolved in 14 mL acetone in a flask. Then 2 mL 30% trimethylamine solution was added quickly. The mixture was stirred and refluxed at 60 °C overnight. After that, the solution was cooled to ambient temperature and acetone was removed by vacuum. After being extracted by pure water and dichloromethane twice, the aqueous phase was collected and concentrated. The solid was redissolved by 1 mL methanol and precipitated by adding 50 mL diethyl ether. Finally, white powder (65 mg, yield 80%) was acquired by centrifuging.
1H-NMR (600 MHz, DMSO-d6, δ): 9.27 (s, 1H), 8.37 (s, 1H), 8.34 (s, 2H), 8.30 (s, 2H), 8.20 (s, 2H), 8.10 (s, 2H), 5.10 (s, 2H), 4.24 (s, 2H), 3.27 (s, 9H).
RESULTS AND DISCUSSION
Temperature Response of the Polymer Fluorescent System
To investigate the temperature response behavior of the system, we adopted 20 μmol/L Py-N and 50 μmol/L PSS for the system. Figure 1(b) demonstrates the absorption spectra of Py-N and PSS system at different temperatures. Two absorption bands around 275 nm and 350 nm were observed respectively. It could be seen that the two absorption bands were not temperature-dependent over the temperature range of 10-70 °C. In contrast, the fluorescence spectra of the polymer system at various temperatures showed great difference. For the system of Py-N and PSS in PBS buffer (as shown in Fig. 1c), when temperature was kept in 10 °C, the system exhibited stronger excimer emission and weaker monomer emission compared with those at room temperature. As temperature rose, the excimer emission intensity decreased gradually, while the monomer emission intensity increased at the same time. At 68 °C, excimer emission almost disappeared, and only monomer emission could be clearly observed. These results confirmed our proposed response mechanism, namely the electrostatic interaction induced the aggregation of Py-N and PSS, which accordingly resulted in excimer emission; while at higher temperatures, deaggregation led to monomer emission. Furthermore, as the temperature rises, the quantum yields of this aqueous fluorescent system also changes, with the quantum yield of the monomer emission increasing and that of the excimer decreasing, as shown in Table S1. The results indicate that the system is effective for detecting temperature changes with the potential for acting as novel fluorescent thermometer.
In addition, the fluorescence intensity ratio (I399/I490) versus temperature curve for the system is displayed in Fig. 1(d). In the relatively high temperature range (40-68 °C), the ratio increased significantly with temperature rise, finally I399/I490 reached 18. This curve can be fitted by a fifth-order polynomial (the equation above Fig. 1d, where I’ represents I399/I490, R2 = 0.9997).
To further demonstrate the excimer/monomer fluorescence interconversion of the system was induced by the synergistic effect of temperature and electrostatic interaction, a control experiment for Py-N solution without PSS was carried out (Fig. S5). When excited at 10 °C, 20 μmol/L Py-N in PBS buffer only showed strong monomer emission between 379-399 nm and it was hard to find any excimer emission around 490 nm. As the temperature rose, the monomer fluorescence of Py-N had a slight increase, still with no excimer emission. Therefore, the results of the control experiment indicate that, Py-N alone cannot act as a fluorescence thermometer, and the electrostatic interaction between Py-N and PSS plays a key role in the monomer/excimer fluorescence conversion of the system.
Preparation of the Polymer Fluorescent System
To prepare the polymer fluorescent thermosensitive system, the positively charged pyrene derivative (Py-N) was synthesized and characterized. The two components of the system (Py-N and PSS) were dissolved in PBS buffer (pH 7.4). In order to ensure optimal performance of the fluorescent thermometer system, it is crucial to determine a suitable ratio of Py-N and PSS. Hence we first investigated the fluorescence spectra of Py-N (20 μmol/L) in PBS buffer (pH 7.4) with various concentrations of PSS, as shown in Fig. 1(a). In the absence of PSS in the solution, only the emission with double peaks at 379-399 nm (monomer emission) could be observed. Upon addition of PSS, a new broad emission band appeared around 490 nm, representing the excimer emission of Py-N. In addition, the excimer emission of Py-N increased in a gradual way, and in the meantime the monomer emission decreased gradually along with the increase of PSS. The excimer emission of Py-N reflects the aggregation of Py-N molecules around the negatively charged PSS polymer chain. The more PSS was added, the more Py-N molecules could bind along the polymer chain via electrostatic-interaction-mediated aggregation. But it is also found that when the amount of PSS exceeded 100 μmol/L, the monomer emission increased again, probably because the excess polyelectrolyte lowered the aggregation extent of Py-N molecules (Fig S4). It can also be observed that, when the amount of PSS exceeds 40 μmol/L, quite strong excimer emission can be observed, which would be helpful for achieving better performance of fluorescence conversion. Based on the results, it is determined that, the appropriate amount of PSS is in the range of 40-100 μmol/L for 20 μmol/L Py-N.
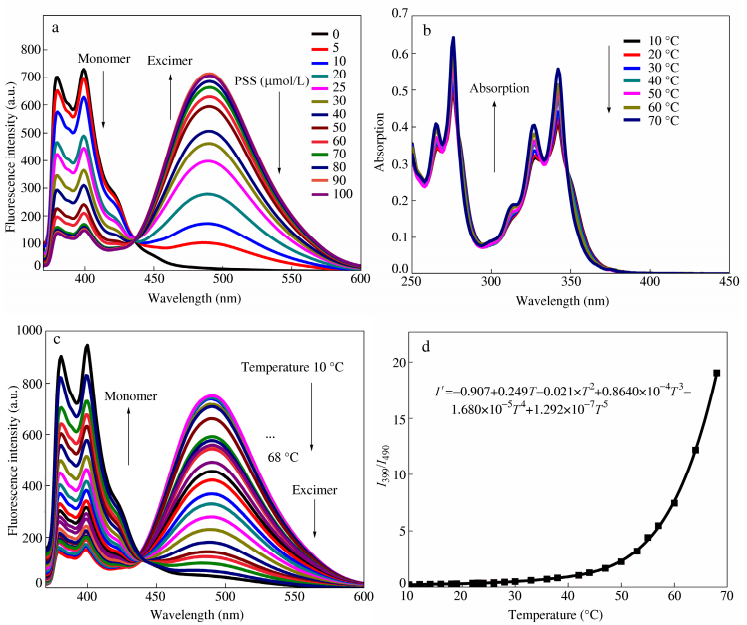 Figure 1.
(a) Fluorescence spectra of Py-N (20 μmol/L) in the presence of different amounts of PSS at 25 °C; (b) Absorption spectra of 20 μmol/L Py-N and 50 μmol/L PSS at different temperatures; (c) Fluorescence spectra of 20 μmol/L Py-N and 50 μmol/L PSS at different temperatures and (d) Fluorescence intensity ratio (I399/I490) versus temperature (Excitation wavelength: 350 nm, 50 mmol/L PBS buffer (pH 7.4))
Figure 1.
(a) Fluorescence spectra of Py-N (20 μmol/L) in the presence of different amounts of PSS at 25 °C; (b) Absorption spectra of 20 μmol/L Py-N and 50 μmol/L PSS at different temperatures; (c) Fluorescence spectra of 20 μmol/L Py-N and 50 μmol/L PSS at different temperatures and (d) Fluorescence intensity ratio (I399/I490) versus temperature (Excitation wavelength: 350 nm, 50 mmol/L PBS buffer (pH 7.4))
Applications of the Polymer Fluorescent System
To explore the applications of the system, we further evaluated this temperature-dependent system, and employed the fluorescence spectra data to obtain the Commission Internationale de L’Eclairage (CIE) 1931 coordinates. The CIE utilizes mathematical method to describe color and is device independent[27]. The color change of the fluorescence in the CIE (x, y) chromaticity diagram at different temperatures for the system is shown in Fig. 2(a). It can be easily observed the variation range (from blue-violet to blue-green) of fluorescence caused by temperature rise. Moreover, the heating/cooling cycle experiment (Fig. 2b) showed that the system of 20 μmol/L Py-N and 50 μmol/L PSS in PBS buffer had quite good fluorescence repeatability and thermo-responsive reversibility.
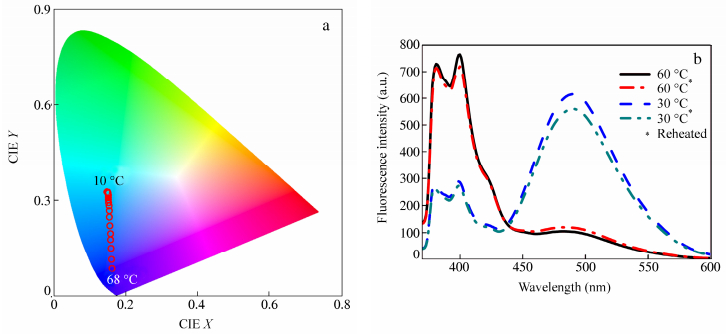 Figure 2.
(a) CIE chromaticity diagram showing the temperature dependence of the (x, y) color coordinates of the system of 20 μmol/L Py-N and 50 μmol/L PSS in PBS buffer; (b) Fluorescence spectra for 20 μmol/L Py-N and 50 μmol/L PSS in PBS buffer (The system was reheated after natural cooling to room temperature, and the fluorescence spectra were measured once the solution was heated or cooled to the set temperature.)
Figure 2.
(a) CIE chromaticity diagram showing the temperature dependence of the (x, y) color coordinates of the system of 20 μmol/L Py-N and 50 μmol/L PSS in PBS buffer; (b) Fluorescence spectra for 20 μmol/L Py-N and 50 μmol/L PSS in PBS buffer (The system was reheated after natural cooling to room temperature, and the fluorescence spectra were measured once the solution was heated or cooled to the set temperature.)
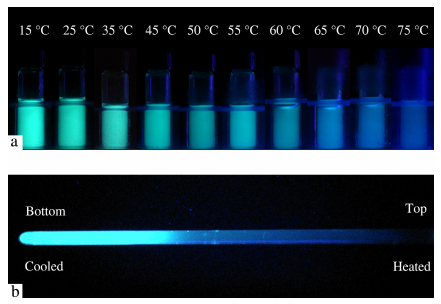
To demonstrate the application of polymer fluorescent system, the photographs for fluorescence changes resulted from temperature changes are shown in Fig. 3(a). The solutions (20 μmol/L Py-N and 50 μmol/L PSS in PBS buffer) were sealed into transparent vials, heated by water bath and excited under a 365 nm hand-held UV lamp. Obviously the system of Py-N and PSS in PBS buffer exhibited quite good performance with a relatively wide color change (from 15-75 °C), and the results were easily observed by naked eyes.
Considering that some precision instruments or therapeutic systems are required to monitor their operating temperature and temperature distribution, we have tried to demonstrate the application of our polymer fluorescent system under a temperature gradient condition. As shown in Fig. 3(b), 2 mL of the solution of 20 μmol/L Py-N and 50 μmol/L PSS in PBS buffer was put into a quartz tube, and the tube was cooled by ice water from the bottom and heated by a blow drier from the top. In this case, the tube formed a vertical temperature gradient and displayed obvious fluorescence change along the temperature gradient. At the bottom of the tube, bright green fluorescence could be observed, and from the bottom to the top, the fluorescence color gradually changed to blue, and finally the heated part exhibited weak violet. The fluorescent changes are mainly in conformity with the CIE diagram. This result shows that the polymer fluorescent system has the potential for being applied in detecting temperature and temperature distribution, through direct observation by naked eyes or a CCD camera.
CONCLUSIONS
In summary, based on excimer/monomer interconversion of charged pyrene derivative and electrostatic interaction mediated aggregation/deaggregation, a facile and effective polymer fluorescent thermosensitive system was successfully developed. The system exhibited quite good excimer/monomer conversion, thermosensitive feature and thermo-responsive reversibility. More importantly, by using this system as a thermometer, fluorescence changes resulted from temperature or temperature distribution changes could be easily observed and was proven to be in conformity with the CIE diagram. Therefore, this thermosensitive polymer fluorescent system shows great potential for serving as a fluorescent thermometer in complex environment.
-
-
[1]
Yang, Z.G., Cao, J.F., He, Y.X., Yang, J.H., Kim, T., Peng, X.J. and Kim, J.S., Chem. Soc. Rev., 2014, 43: 4563
-
[2]
Wang, Z.Y., Ma, X.Q., Zong, S.F., Wang, Y.Z., Chen, H. and Cui, Y.P., Talanta, 2015, 131: 259
-
[3]
Pais, V.F., Lassaletta, J.M., Fernandez, R., El-Sheshtawy, H.S., Ros, A. and Pischel, U., Chem. Eur. J., 2014, 20: 7638
-
[4]
Feng, J., Xiong, L., Wang, S.Q., Li, S.Y., Li, Y. and Yang, G.Q., Adv. Funct. Mater., 2013, 23: 340
-
[5]
Ozawa, A., Shimizu, A., Nishiyabu, R. and Kubo, Y., Chem. Commun., 2015, 51: 118
-
[6]
Baker, G.A., Baker, S.N. and McCleskey, T.M., Chem. Commun., 2003, 23: 2932
-
[7]
Hang, Y.D., He, X.P., Yang, L. and Hua, J.L., Biosens. Bioelectron., 2015, 65: 420
-
[8]
Ji, X.F., Wang, P., Wang, H. and Huang, F.H., Chinese J. Polym. Sci., 2015, 33(6): 890
-
[9]
Zhang, X.Q., Zhang, X.Y., Yang, B. and Wei, Y., Chinese J. Polym. Sci., 2014, 32(11): 1479
-
[10]
Huang, Y.F., Ou, D.X., Wang, C., Huang, C., Li, Q.Q. and Li, Z., Polym. Chem., 2014, 5: 2041
-
[11]
Qiu, T., Chen, Y., Song, J. and Fan, L.J., ACS Appl. Mater. Interfaces, 2015, 7: 8260
-
[12]
Xu, X.Q., Miao, K.S., Chen, Y. and Fan, L.J., ACS Appl. Mater. Interfaces, 2015, 7: 7759
-
[13]
Gao, Y.T., Feng, G.X., Jiang, T., Goh, C.C., Ng, L.G., Liu, B., Li, B., Yang, L., Hua, J.L., and Tian, H., Adv. Funct. Mater., 2015, 25: 2857
-
[14]
Huang, J., Sun, N., Yang, J., Tang, R.L., Li, Q.Q., Ma, D.G. and Li, Z., Adv. Funct. Mater. 2014, 24, 7645
-
[15]
Lee, J. and Kotov, N.A., Nano Today, 2007, 2(1): 48
-
[16]
Li, J., Jiang, Y.B., Cheng, J., Zhang, Y.L., Su, H.M., Lam, J.W.Y., Sung, H.H.Y., Wong, K.S., Kwok, H.S. and Tang, B.Z., Phys. Chem. Chem. Phys., 2015: 17, 1134
-
[17]
Ebrahimi, S., Akhlaghi, Y., Kompany-Zareh, M. and Rinnan, A., ACS Nano, 2014, 8(10): 10372
-
[18]
Wang, X.D., Meier, R.J., Schmittlein, C., Schreml, S., Schaferling, M. and Wolfbeis, O.S., Sensor. Actuat. B-Chem., 2015, 221: 37
-
[19]
Liu, G.F., Zhou, W., Zhang, J.Q. and Zhao, P., J. Polym. Sci., Part A: Polym. Chem., 2012, 50: 2219
-
[20]
Liu, Y., Ma, C., Zeng, F. and Wu, S.Z., Acta Polymerica Sinica (in Chinese), 2012, (6): 666
-
[21]
Uchiyama, S., Matsumura, Y., de Silva, A.P. and Iwai, K., Anal. Chem., 2004, 76: 1793
-
[22]
Tsuji, T., Yoshida, S., Yoshida, A. and Uchiyama, S., Anal. Chem., 2013, 85: 9815
-
[23]
Jiang, Y.N., Yang, X.D., Ma, C., Wang, C.X., Chen, Y., Dong, F.X., Yang, B., Yu, K. and Lin, Q., ACS Appl. Mater. Interfaces, 2014, 6: 4650
-
[24]
Wang, X., Guo, X.H., Zhu, Y., Li, L., Wu, S. and Zhang, R., Chinese J. Polym. Sci., 2011, 29(4): 490
-
[25]
Qiao, J., Chen, C.F., Qi, L., Liu, M.R., Dong, P., Jiang, Q., Yang, X.Z., Mu, X.Y. and Mao, L.Q., J. Mater. Chem. B, 2014, 2: 7544
-
[26]
Lee, S., Lee, J.S., Lee, C.H., Jung, Y.S. and Kim, J.M., Langmuir, 2011, 27(5): 1560
-
[27]
Feng, J., Tian, K.J., Hu, D.H., Wang, S.Q., Li, S.Y., Zeng, Y., Li, Y. and Yang, G.Q., Angew. Chem. Int. Ed., 2011, 50: 8072
-
[28]
Wang, H., Wu, Y.Q., Shi, Y.L., Tao, P., Fan, X., Su, X.Y. and Kuang, G.C., Chem. Eur. J., 2015, 21: 3219
-
[29]
Liu, X., Li, S.Y., Feng, J., Li, Y. and Yang, G.Q., Chem. Commun., 2014, 50: 2778
-
[30]
Cao, C., Liu, X.G., Qiao, Q.L., Zhao, M., Yin, W.T., Mao, D.Q., Zhang, H. and Xu, Z.C., Chem. Commun., 2014, 50: 15811
-
[31]
Liu, L.X., Li, W., Yan, J.T. and Zhang, A.F., J. Polym. Sci., Part A: Polym. Chem., 2014, 52: 1706
-
[32]
Chen, Y.P. and Li, X.D., Biomacromolecules, 2011, 12: 4367
-
[33]
Wang, C.Y., Tong, Z., Zeng, F., Ren, B.Y. and Liu, X.X., Acta Polymerica Sinica (in Chinese), 2002, (6): 729
-
[34]
Wang, C.Y., Sun, Q.L., Tong, Z., Liu, X.X., Zeng, F. and Gao, F., Acta Polymerica Sinica (in Chinese), 2011, (7): 1265
-
[35]
Hong, S.W., Kim, D.Y., Lee, J.U. and Jo, W.H., Macromolecules, 2009, 42(7): 2009
-
[36]
Zhang, Q.E., Deng, T., Li, J.S., Xu, W.J., Shen, G.L. and Yu, R.Q., Biosens. Bioelectron., 2015, 68: 253
-
[37]
Bibi, I. and Siddiq, M., Chinese J. Polym. Sci., 2011, 29(5): 575
-
[38]
Qiao, J.J., Zhang, X.H. and Wu, S.K., Acta Polymerica Sinica (in Chinese), 2006, (1): 76
-
[39]
Zhegalova, N.G., Dergunov, S.A., Wang, S.T., Pinkhassik, E. and Berezin, M.Y., Chem. Eur. J., 2014, 20: 10292
-
[40]
Takei, Y., Arai, S., Murata, A., Takabayashi, M., Oyama, K., Ishiwata, S., Takeoka, S. and Suzuki, M., ACS Nano, 2014, 8(1): 198
-
[41]
Wu, Y.X., Zhang, X.B., Li, J.B., Zhang, C.C., Liang, H., Mao, G.J., Zhou, L.Y., Tan, W.H., Yu, R.Q., Anal. Chem., 2014, 86: 10389
-
[42]
Wang, F., Nandhakumar, R., Moon, J.H., Kim, K.M., Lee, J.Y. and Yoon, J., Inorg. Chem., 2011, 50: 2240
-
[43]
Chen, H., Lin, W.Y., Jiang, W.Q., Dong, B.L., Cui, H.J. and Tang, Y.H., Chem. Commun., 2015, 51: 6968
-
[44]
Williams, A.T.R., Winfield, S.A. and Miller, J.N., Analyst, 1983, 108: 1067
-
[45]
Demas, J.N. and Crosby, G.A., J. Phys. Chem., 1971, 75 (8): 991
-
[46]
Broadbent, A.D., Color Res. Appl., 2004, 29(4): 26
-
[1]
-

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 0
- 文章访问数: 1837
- HTML全文浏览量: 35

 下载:
下载:

